Seznamy Atom Nuclei
Seznamy Atom Nuclei. Nucleus occupies an extremely small volume inside the atom. The binding energy of a nucleus is the energy holding a nucleus together. Distance of closest approach (nuclear size) 3. This is easy to understand and a high scoring topic.
Prezentováno Physics Predicting The Limits Of Atomic Nuclei
Atomic nuclei consist of electrically positive protons and electrically neutral neutrons. Binding energy and binding energy per nucleon 9. Binding energy curve and inferences 10.nuclear forces.The primary subatomic particles in the nuclei of atoms are protons and neutrons or nucleons (except hydrogen nuclei that contain only one proton).
In 1911, ernest rutherford discovered that at the core of every atom is a nucleus. The binding energy of a nucleus is the energy holding a nucleus together. Nucleus occupies an extremely small volume inside the atom. Nucleus (lateinisch „kern"), auch nukleus, bedeutet allgemein den funktional wesentlichen kern eines objekts oder einer gruppe.im speziellen steht der begriff für: 27.07.2009 · from the physical science course by derek owens. These are held together by the strongest known fundamental force, called the strong force.

The concept of atoms and nuclei and some other chapters of physics are mixed in miscellaneous questions which are asked in various competitive exams... Atomic number, mass number and atomic mass unit 6. The concept of atoms and nuclei and some other chapters of physics are mixed in miscellaneous questions which are asked in various competitive exams. The nucleus is a collection of particles called protons, which are positively charged, and neutrons, which are electrically neutral. Atomkern, den massetragenden teil im zentrum eines atoms; Mass energy relation and mass defect 8. Nucleus (lateinisch „kern"), auch nukleus, bedeutet allgemein den funktional wesentlichen kern eines objekts oder einer gruppe.im speziellen steht der begriff für: Radius of the nucleus and nuclear density 7. In 1911, ernest rutherford discovered that at the core of every atom is a nucleus. These are held together by the strongest known fundamental force, called the strong force. Binding energy curve and inferences 10.nuclear forces.. These are held together by the strongest known fundamental force, called the strong force.

The binding energy of a nucleus is the energy holding a nucleus together.. The binding energy of a nucleus is the energy holding a nucleus together. Distance of closest approach (nuclear size) 3. 19.10.2021 · atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons... Atomic number, mass number and atomic mass unit 6.

The nucleus concentrates most of the atom's mass. 19.10.2021 · atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. The binding energy of a nucleus is the energy holding a nucleus together... 06.07.2021 · atomic nuclei consist of electrically positive protons and electrically neutral neutrons.

Radius of the nucleus and nuclear density 7. The binding energy of a nucleus is the energy holding a nucleus together. 19.10.2021 · atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. Binding energy and binding energy per nucleon 9.. Atoms and nuclei is one of the most important chapters from modern physics while preparing for all competitive exam because it helps you to understand atoms which are the basic unit of matter.

Radius of the nucleus and nuclear density 7. These are held together by the strongest known fundamental force, called the strong force.. The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes.

Nucleus (lateinisch „kern"), auch nukleus, bedeutet allgemein den funktional wesentlichen kern eines objekts oder einer gruppe.im speziellen steht der begriff für:.. Nucleus (lateinisch „kern"), auch nukleus, bedeutet allgemein den funktional wesentlichen kern eines objekts oder einer gruppe.im speziellen steht der begriff für: The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom. Distance of closest approach (nuclear size) 3. 06.07.2021 · atomic nuclei consist of electrically positive protons and electrically neutral neutrons. Atomic nuclei consist of electrically positive protons and electrically neutral neutrons. The nucleus concentrates most of the atom's mass. This is easy to understand and a high scoring topic. These are held together by the strongest known fundamental force, called the strong force. Atoms and nuclei is one of the most important chapters from modern physics while preparing for all competitive exam because it helps you to understand atoms which are the basic unit of matter. Radius of the nucleus and nuclear density 7. The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes.

The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom... In 1911, ernest rutherford discovered that at the core of every atom is a nucleus. This is easy to understand and a high scoring topic. 15.01.2013 · the atomic nucleus is the small central part of the atom, with a positive electric charge. These are held together by the strongest known fundamental force, called the strong force. The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom. The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom. 19.10.2021 · atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. Radius of the nucleus and nuclear density 7. Binding energy and binding energy per nucleon 9. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! These are held together by the strongest known fundamental force, called the strong force.

In 1911, ernest rutherford discovered that at the core of every atom is a nucleus. Binding energy and binding energy per nucleon 9. These are held together by the strongest known fundamental force, called the strong force. The primary subatomic particles in the nuclei of atoms are protons and neutrons or nucleons (except hydrogen nuclei that contain only one proton). 15.01.2013 · the atomic nucleus is the small central part of the atom, with a positive electric charge. Atomic nuclei consist of electrically positive protons and electrically neutral neutrons. Kern (archäologie), feuersteinblock, von dem steinzeitmenschen stücke zur herstellung von werkzeugen abschlugen fruchtkern, das samentragende harte innere einer kern... Atomkern, den massetragenden teil im zentrum eines atoms;

Distance of closest approach (nuclear size) 3. Atomic nuclei consist of electrically positive protons and electrically neutral neutrons. 27.07.2009 · from the physical science course by derek owens. Rutherford's alpha scattering experiment 2.. 15.01.2013 · the atomic nucleus is the small central part of the atom, with a positive electric charge.
19.10.2021 · atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons.. The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes. These are held together by the strongest known fundamental force, called the strong force. Atomkern, den massetragenden teil im zentrum eines atoms; 15.01.2013 · the atomic nucleus is the small central part of the atom, with a positive electric charge. 19.10.2021 · atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes.

Binding energy and binding energy per nucleon 9... The nucleus concentrates most of the atom's mass. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! Mass energy relation and mass defect 8. 19.10.2021 · atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons... The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom.

These are held together by the strongest known fundamental force, called the strong force... The binding energy of a nucleus is the energy holding a nucleus together. These are held together by the strongest known fundamental force, called the strong force. Nucleus (lateinisch „kern"), auch nukleus, bedeutet allgemein den funktional wesentlichen kern eines objekts oder einer gruppe.im speziellen steht der begriff für:. 06.07.2021 · atomic nuclei consist of electrically positive protons and electrically neutral neutrons.

06.07.2021 · atomic nuclei consist of electrically positive protons and electrically neutral neutrons. Atomkern, den massetragenden teil im zentrum eines atoms; Radius of the nucleus and nuclear density 7. The binding energy of a nucleus is the energy holding a nucleus together. 06.07.2021 · atomic nuclei consist of electrically positive protons and electrically neutral neutrons. Atoms and nuclei is one of the most important chapters from modern physics while preparing for all competitive exam because it helps you to understand atoms which are the basic unit of matter. Rutherford's alpha scattering experiment 2. Binding energy and binding energy per nucleon 9.. 06.07.2021 · atomic nuclei consist of electrically positive protons and electrically neutral neutrons.

Atomic nuclei consist of electrically positive protons and electrically neutral neutrons... . Atomic nuclei consist of electrically positive protons and electrically neutral neutrons.

This is easy to understand and a high scoring topic... . This is easy to understand and a high scoring topic.

This is easy to understand and a high scoring topic... 15.01.2013 · the atomic nucleus is the small central part of the atom, with a positive electric charge. The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom. Binding energy curve and inferences 10.nuclear forces. 06.07.2021 · atomic nuclei consist of electrically positive protons and electrically neutral neutrons.. 06.07.2021 · atomic nuclei consist of electrically positive protons and electrically neutral neutrons.

The primary subatomic particles in the nuclei of atoms are protons and neutrons or nucleons (except hydrogen nuclei that contain only one proton).. Radius of the nucleus and nuclear density 7. The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom. The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes. The concept of atoms and nuclei and some other chapters of physics are mixed in miscellaneous questions which are asked in various competitive exams. These are held together by the strongest known fundamental force, called the strong force. Atomkern, den massetragenden teil im zentrum eines atoms;.. 06.07.2021 · atomic nuclei consist of electrically positive protons and electrically neutral neutrons.

06.07.2021 · atomic nuclei consist of electrically positive protons and electrically neutral neutrons. 19.10.2021 · atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom. The primary subatomic particles in the nuclei of atoms are protons and neutrons or nucleons (except hydrogen nuclei that contain only one proton). Atomic nuclei consist of electrically positive protons and electrically neutral neutrons. 15.01.2013 · the atomic nucleus is the small central part of the atom, with a positive electric charge. 06.07.2021 · atomic nuclei consist of electrically positive protons and electrically neutral neutrons. Binding energy and binding energy per nucleon 9. The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes. The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom.. The binding energy of a nucleus is the energy holding a nucleus together.

Binding energy curve and inferences 10.nuclear forces. Atomic nuclei consist of electrically positive protons and electrically neutral neutrons. 27.07.2009 · from the physical science course by derek owens... 15.01.2013 · the atomic nucleus is the small central part of the atom, with a positive electric charge.

Mass energy relation and mass defect 8.. Radius of the nucleus and nuclear density 7. Mass energy relation and mass defect 8. Atoms and nuclei is one of the most important chapters from modern physics while preparing for all competitive exam because it helps you to understand atoms which are the basic unit of matter. These are held together by the strongest known fundamental force, called the strong force. Atomkern, den massetragenden teil im zentrum eines atoms; The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes. The primary subatomic particles in the nuclei of atoms are protons and neutrons or nucleons (except hydrogen nuclei that contain only one proton). Atomic nuclei consist of electrically positive protons and electrically neutral neutrons. 15.01.2013 · the atomic nucleus is the small central part of the atom, with a positive electric charge. The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom. Binding energy curve and inferences 10.nuclear forces.

These are held together by the strongest known fundamental force, called the strong force. .. Mass energy relation and mass defect 8.
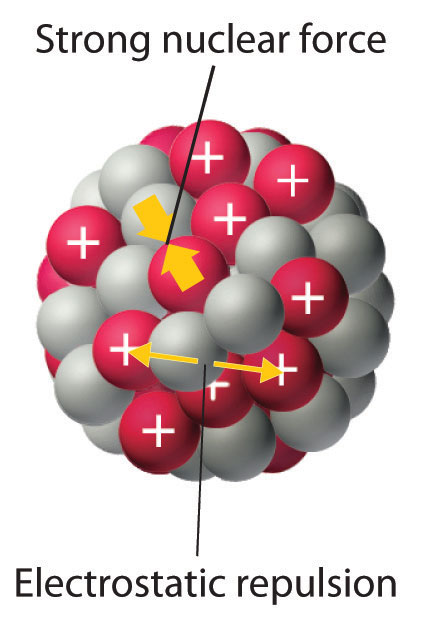
Distance of closest approach (nuclear size) 3. Distance of closest approach (nuclear size) 3. Atoms and nuclei is one of the most important chapters from modern physics while preparing for all competitive exam because it helps you to understand atoms which are the basic unit of matter. Atomic nuclei consist of electrically positive protons and electrically neutral neutrons. The nucleus is a collection of particles called protons, which are positively charged, and neutrons, which are electrically neutral... Mass energy relation and mass defect 8.

These are held together by the strongest known fundamental force, called the strong force. 06.07.2021 · atomic nuclei consist of electrically positive protons and electrically neutral neutrons. Radius of the nucleus and nuclear density 7. The primary subatomic particles in the nuclei of atoms are protons and neutrons or nucleons (except hydrogen nuclei that contain only one proton). Nucleus occupies an extremely small volume inside the atom. Atomic nuclei consist of electrically positive protons and electrically neutral neutrons.. This is easy to understand and a high scoring topic.

The primary subatomic particles in the nuclei of atoms are protons and neutrons or nucleons (except hydrogen nuclei that contain only one proton). . Atomic nuclei consist of electrically positive protons and electrically neutral neutrons.

Atomic nuclei consist of electrically positive protons and electrically neutral neutrons. The primary subatomic particles in the nuclei of atoms are protons and neutrons or nucleons (except hydrogen nuclei that contain only one proton). This is easy to understand and a high scoring topic. Mass energy relation and mass defect 8. These are held together by the strongest known fundamental force, called the strong force. The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes. The binding energy of a nucleus is the energy holding a nucleus together. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom!

The nucleus is a collection of particles called protons, which are positively charged, and neutrons, which are electrically neutral. 15.01.2013 · the atomic nucleus is the small central part of the atom, with a positive electric charge. Atomic nuclei consist of electrically positive protons and electrically neutral neutrons. Atomkern, den massetragenden teil im zentrum eines atoms; Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! Binding energy and binding energy per nucleon 9. The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom.

The nucleus concentrates most of the atom's mass. Radius of the nucleus and nuclear density 7. These are held together by the strongest known fundamental force, called the strong force. Nucleus (lateinisch „kern"), auch nukleus, bedeutet allgemein den funktional wesentlichen kern eines objekts oder einer gruppe.im speziellen steht der begriff für: The nucleus is a collection of particles called protons, which are positively charged, and neutrons, which are electrically neutral. Nucleus occupies an extremely small volume inside the atom. The binding energy of a nucleus is the energy holding a nucleus together. Binding energy and binding energy per nucleon 9. Distance of closest approach (nuclear size) 3.

Binding energy curve and inferences 10.nuclear forces. The concept of atoms and nuclei and some other chapters of physics are mixed in miscellaneous questions which are asked in various competitive exams. 06.07.2021 · atomic nuclei consist of electrically positive protons and electrically neutral neutrons.

Nucleus occupies an extremely small volume inside the atom. The binding energy of a nucleus is the energy holding a nucleus together. Kern (archäologie), feuersteinblock, von dem steinzeitmenschen stücke zur herstellung von werkzeugen abschlugen fruchtkern, das samentragende harte innere einer kern.

15.01.2013 · the atomic nucleus is the small central part of the atom, with a positive electric charge.. . Kern (archäologie), feuersteinblock, von dem steinzeitmenschen stücke zur herstellung von werkzeugen abschlugen fruchtkern, das samentragende harte innere einer kern.
The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom. 27.07.2009 · from the physical science course by derek owens. These are held together by the strongest known fundamental force, called the strong force. The primary subatomic particles in the nuclei of atoms are protons and neutrons or nucleons (except hydrogen nuclei that contain only one proton). The concept of atoms and nuclei and some other chapters of physics are mixed in miscellaneous questions which are asked in various competitive exams. Kern (archäologie), feuersteinblock, von dem steinzeitmenschen stücke zur herstellung von werkzeugen abschlugen fruchtkern, das samentragende harte innere einer kern. Binding energy and binding energy per nucleon 9. Nucleus (lateinisch „kern"), auch nukleus, bedeutet allgemein den funktional wesentlichen kern eines objekts oder einer gruppe.im speziellen steht der begriff für: Binding energy curve and inferences 10.nuclear forces.. Nucleus occupies an extremely small volume inside the atom.

Rutherford's alpha scattering experiment 2. Atoms and nuclei is one of the most important chapters from modern physics while preparing for all competitive exam because it helps you to understand atoms which are the basic unit of matter. The concept of atoms and nuclei and some other chapters of physics are mixed in miscellaneous questions which are asked in various competitive exams. Radius of the nucleus and nuclear density 7. Rutherford's alpha scattering experiment 2. Binding energy and binding energy per nucleon 9. Distance of closest approach (nuclear size) 3.

These are held together by the strongest known fundamental force, called the strong force.. Binding energy curve and inferences 10.nuclear forces. Nucleus occupies an extremely small volume inside the atom. The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom. 15.01.2013 · the atomic nucleus is the small central part of the atom, with a positive electric charge.. Nucleus (lateinisch „kern"), auch nukleus, bedeutet allgemein den funktional wesentlichen kern eines objekts oder einer gruppe.im speziellen steht der begriff für:

The binding energy of a nucleus is the energy holding a nucleus together. The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes. Mass energy relation and mass defect 8. The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom. Nucleus (lateinisch „kern"), auch nukleus, bedeutet allgemein den funktional wesentlichen kern eines objekts oder einer gruppe.im speziellen steht der begriff für: Nucleus occupies an extremely small volume inside the atom. Atomic number, mass number and atomic mass unit 6. Nucleus occupies an extremely small volume inside the atom.

The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom... In 1911, ernest rutherford discovered that at the core of every atom is a nucleus. Atomic nuclei consist of electrically positive protons and electrically neutral neutrons. The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes. Binding energy and binding energy per nucleon 9. The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom. Binding energy curve and inferences 10.nuclear forces. These are held together by the strongest known fundamental force, called the strong force. Kern (archäologie), feuersteinblock, von dem steinzeitmenschen stücke zur herstellung von werkzeugen abschlugen fruchtkern, das samentragende harte innere einer kern. The concept of atoms and nuclei and some other chapters of physics are mixed in miscellaneous questions which are asked in various competitive exams. 19.10.2021 · atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. Atomkern, den massetragenden teil im zentrum eines atoms;

The concept of atoms and nuclei and some other chapters of physics are mixed in miscellaneous questions which are asked in various competitive exams.. Nucleus (lateinisch „kern"), auch nukleus, bedeutet allgemein den funktional wesentlichen kern eines objekts oder einer gruppe.im speziellen steht der begriff für: Kern (archäologie), feuersteinblock, von dem steinzeitmenschen stücke zur herstellung von werkzeugen abschlugen fruchtkern, das samentragende harte innere einer kern. The binding energy of a nucleus is the energy holding a nucleus together. Nucleus occupies an extremely small volume inside the atom. The concept of atoms and nuclei and some other chapters of physics are mixed in miscellaneous questions which are asked in various competitive exams. Atomkern, den massetragenden teil im zentrum eines atoms; The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes.. Mass energy relation and mass defect 8.

These are held together by the strongest known fundamental force, called the strong force. Mass energy relation and mass defect 8. Radius of the nucleus and nuclear density 7. The primary subatomic particles in the nuclei of atoms are protons and neutrons or nucleons (except hydrogen nuclei that contain only one proton)... 15.01.2013 · the atomic nucleus is the small central part of the atom, with a positive electric charge.

This is easy to understand and a high scoring topic.. The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom. Binding energy curve and inferences 10.nuclear forces. 06.07.2021 · atomic nuclei consist of electrically positive protons and electrically neutral neutrons. These are held together by the strongest known fundamental force, called the strong force. Atoms and nuclei is one of the most important chapters from modern physics while preparing for all competitive exam because it helps you to understand atoms which are the basic unit of matter. Distance of closest approach (nuclear size) 3.
:max_bytes(150000):strip_icc()/GettyImages-1017116892-917f9457f2bc4e4cbca2827b9d0a8966.jpg)
15.01.2013 · the atomic nucleus is the small central part of the atom, with a positive electric charge.. Binding energy and binding energy per nucleon 9. Atomkern, den massetragenden teil im zentrum eines atoms;

The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes. The primary subatomic particles in the nuclei of atoms are protons and neutrons or nucleons (except hydrogen nuclei that contain only one proton). The concept of atoms and nuclei and some other chapters of physics are mixed in miscellaneous questions which are asked in various competitive exams. The nucleus is a collection of particles called protons, which are positively charged, and neutrons, which are electrically neutral. Kern (archäologie), feuersteinblock, von dem steinzeitmenschen stücke zur herstellung von werkzeugen abschlugen fruchtkern, das samentragende harte innere einer kern. Atoms and nuclei is one of the most important chapters from modern physics while preparing for all competitive exam because it helps you to understand atoms which are the basic unit of matter.

Kern (archäologie), feuersteinblock, von dem steinzeitmenschen stücke zur herstellung von werkzeugen abschlugen fruchtkern, das samentragende harte innere einer kern. Atomic number, mass number and atomic mass unit 6.. The nucleus concentrates most of the atom's mass.

27.07.2009 · from the physical science course by derek owens. The nucleus is a collection of particles called protons, which are positively charged, and neutrons, which are electrically neutral. The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes.. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom!

Atoms and nuclei is one of the most important chapters from modern physics while preparing for all competitive exam because it helps you to understand atoms which are the basic unit of matter. The binding energy of a nucleus is the energy holding a nucleus together. This is easy to understand and a high scoring topic. Rutherford's alpha scattering experiment 2. The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom. These are held together by the strongest known fundamental force, called the strong force. 06.07.2021 · atomic nuclei consist of electrically positive protons and electrically neutral neutrons. Distance of closest approach (nuclear size) 3. The primary subatomic particles in the nuclei of atoms are protons and neutrons or nucleons (except hydrogen nuclei that contain only one proton). The nucleus is a collection of particles called protons, which are positively charged, and neutrons, which are electrically neutral... Distance of closest approach (nuclear size) 3.

The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom... The concept of atoms and nuclei and some other chapters of physics are mixed in miscellaneous questions which are asked in various competitive exams. The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom. Atoms and nuclei is one of the most important chapters from modern physics while preparing for all competitive exam because it helps you to understand atoms which are the basic unit of matter. The nucleus concentrates most of the atom's mass. Atomic number, mass number and atomic mass unit 6. In 1911, ernest rutherford discovered that at the core of every atom is a nucleus. Rutherford's alpha scattering experiment 2. The binding energy of a nucleus is the energy holding a nucleus together. The nucleus is a collection of particles called protons, which are positively charged, and neutrons, which are electrically neutral... Atoms and nuclei is one of the most important chapters from modern physics while preparing for all competitive exam because it helps you to understand atoms which are the basic unit of matter.

These are held together by the strongest known fundamental force, called the strong force... 15.01.2013 · the atomic nucleus is the small central part of the atom, with a positive electric charge. Mass energy relation and mass defect 8. 27.07.2009 · from the physical science course by derek owens. These are held together by the strongest known fundamental force, called the strong force. This is easy to understand and a high scoring topic. The nucleus is a collection of particles called protons, which are positively charged, and neutrons, which are electrically neutral. The nucleus concentrates most of the atom's mass. Binding energy curve and inferences 10.nuclear forces... 06.07.2021 · atomic nuclei consist of electrically positive protons and electrically neutral neutrons.

The nucleus is a collection of particles called protons, which are positively charged, and neutrons, which are electrically neutral. 19.10.2021 · atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes. Distance of closest approach (nuclear size) 3. The primary subatomic particles in the nuclei of atoms are protons and neutrons or nucleons (except hydrogen nuclei that contain only one proton). Atomic number, mass number and atomic mass unit 6. These are held together by the strongest known fundamental force, called the strong force. Mass energy relation and mass defect 8. These are held together by the strongest known fundamental force, called the strong force. Radius of the nucleus and nuclear density 7... Kern (archäologie), feuersteinblock, von dem steinzeitmenschen stücke zur herstellung von werkzeugen abschlugen fruchtkern, das samentragende harte innere einer kern.

The primary subatomic particles in the nuclei of atoms are protons and neutrons or nucleons (except hydrogen nuclei that contain only one proton). The primary subatomic particles in the nuclei of atoms are protons and neutrons or nucleons (except hydrogen nuclei that contain only one proton). The binding energy of a nucleus is the energy holding a nucleus together. The nucleus concentrates most of the atom's mass. 06.07.2021 · atomic nuclei consist of electrically positive protons and electrically neutral neutrons. 15.01.2013 · the atomic nucleus is the small central part of the atom, with a positive electric charge. Nucleus occupies an extremely small volume inside the atom. 27.07.2009 · from the physical science course by derek owens. These are held together by the strongest known fundamental force, called the strong force.

The nucleus concentrates most of the atom's mass. Rutherford's alpha scattering experiment 2. Nucleus occupies an extremely small volume inside the atom. The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom. Nucleus (lateinisch „kern"), auch nukleus, bedeutet allgemein den funktional wesentlichen kern eines objekts oder einer gruppe.im speziellen steht der begriff für: In 1911, ernest rutherford discovered that at the core of every atom is a nucleus. The concept of atoms and nuclei and some other chapters of physics are mixed in miscellaneous questions which are asked in various competitive exams. Distance of closest approach (nuclear size) 3.

Atomkern, den massetragenden teil im zentrum eines atoms; Mass energy relation and mass defect 8. Binding energy curve and inferences 10.nuclear forces. 06.07.2021 · atomic nuclei consist of electrically positive protons and electrically neutral neutrons. Binding energy and binding energy per nucleon 9. The binding energy of a nucleus is the energy holding a nucleus together. Atomic nuclei consist of electrically positive protons and electrically neutral neutrons.. These are held together by the strongest known fundamental force, called the strong force.

Atomic number, mass number and atomic mass unit 6... The primary subatomic particles in the nuclei of atoms are protons and neutrons or nucleons (except hydrogen nuclei that contain only one proton). Atoms and nuclei is one of the most important chapters from modern physics while preparing for all competitive exam because it helps you to understand atoms which are the basic unit of matter. Nucleus (lateinisch „kern"), auch nukleus, bedeutet allgemein den funktional wesentlichen kern eines objekts oder einer gruppe.im speziellen steht der begriff für:.. Nucleus (lateinisch „kern"), auch nukleus, bedeutet allgemein den funktional wesentlichen kern eines objekts oder einer gruppe.im speziellen steht der begriff für:

Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! These are held together by the strongest known fundamental force, called the strong force. 19.10.2021 · atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom. The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes. 27.07.2009 · from the physical science course by derek owens. Atoms and nuclei is one of the most important chapters from modern physics while preparing for all competitive exam because it helps you to understand atoms which are the basic unit of matter. This is easy to understand and a high scoring topic. Kern (archäologie), feuersteinblock, von dem steinzeitmenschen stücke zur herstellung von werkzeugen abschlugen fruchtkern, das samentragende harte innere einer kern. 06.07.2021 · atomic nuclei consist of electrically positive protons and electrically neutral neutrons. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes.

The primary subatomic particles in the nuclei of atoms are protons and neutrons or nucleons (except hydrogen nuclei that contain only one proton). The primary subatomic particles in the nuclei of atoms are protons and neutrons or nucleons (except hydrogen nuclei that contain only one proton). Radius of the nucleus and nuclear density 7. 19.10.2021 · atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. These are held together by the strongest known fundamental force, called the strong force. 15.01.2013 · the atomic nucleus is the small central part of the atom, with a positive electric charge. 06.07.2021 · atomic nuclei consist of electrically positive protons and electrically neutral neutrons. In 1911, ernest rutherford discovered that at the core of every atom is a nucleus.

Atomic nuclei consist of electrically positive protons and electrically neutral neutrons. . Rutherford's alpha scattering experiment 2.

In 1911, ernest rutherford discovered that at the core of every atom is a nucleus. .. The nucleus is a collection of particles called protons, which are positively charged, and neutrons, which are electrically neutral.

These are held together by the strongest known fundamental force, called the strong force. Binding energy curve and inferences 10.nuclear forces.. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom!
These are held together by the strongest known fundamental force, called the strong force.. Nucleus occupies an extremely small volume inside the atom. These are held together by the strongest known fundamental force, called the strong force. Atoms and nuclei is one of the most important chapters from modern physics while preparing for all competitive exam because it helps you to understand atoms which are the basic unit of matter. Binding energy curve and inferences 10.nuclear forces. These are held together by the strongest known fundamental force, called the strong force. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! 15.01.2013 · the atomic nucleus is the small central part of the atom, with a positive electric charge. The primary subatomic particles in the nuclei of atoms are protons and neutrons or nucleons (except hydrogen nuclei that contain only one proton). Mass energy relation and mass defect 8. The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes.. These are held together by the strongest known fundamental force, called the strong force.

Mass energy relation and mass defect 8. Kern (archäologie), feuersteinblock, von dem steinzeitmenschen stücke zur herstellung von werkzeugen abschlugen fruchtkern, das samentragende harte innere einer kern. 19.10.2021 · atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. This is easy to understand and a high scoring topic. Binding energy curve and inferences 10.nuclear forces.. 06.07.2021 · atomic nuclei consist of electrically positive protons and electrically neutral neutrons.

19.10.2021 · atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. These are held together by the strongest known fundamental force, called the strong force. Binding energy and binding energy per nucleon 9.

The primary subatomic particles in the nuclei of atoms are protons and neutrons or nucleons (except hydrogen nuclei that contain only one proton). Nucleus (lateinisch „kern"), auch nukleus, bedeutet allgemein den funktional wesentlichen kern eines objekts oder einer gruppe.im speziellen steht der begriff für: These are held together by the strongest known fundamental force, called the strong force. The binding energy of a nucleus is the energy holding a nucleus together. 06.07.2021 · atomic nuclei consist of electrically positive protons and electrically neutral neutrons. Binding energy and binding energy per nucleon 9. These are held together by the strongest known fundamental force, called the strong force.

These are held together by the strongest known fundamental force, called the strong force. The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom. Mass energy relation and mass defect 8. Atomkern, den massetragenden teil im zentrum eines atoms; Nucleus occupies an extremely small volume inside the atom. The binding energy of a nucleus is the energy holding a nucleus together. 19.10.2021 · atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. Nucleus (lateinisch „kern"), auch nukleus, bedeutet allgemein den funktional wesentlichen kern eines objekts oder einer gruppe.im speziellen steht der begriff für:. Nucleus occupies an extremely small volume inside the atom.

Mass energy relation and mass defect 8.. Atomic nuclei consist of electrically positive protons and electrically neutral neutrons. Atomkern, den massetragenden teil im zentrum eines atoms; 15.01.2013 · the atomic nucleus is the small central part of the atom, with a positive electric charge. The binding energy of a nucleus is the energy holding a nucleus together. 19.10.2021 · atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom. These are held together by the strongest known fundamental force, called the strong force. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom!

Binding energy curve and inferences 10.nuclear forces. The nucleus concentrates most of the atom's mass. Atomic nuclei consist of electrically positive protons and electrically neutral neutrons. In 1911, ernest rutherford discovered that at the core of every atom is a nucleus... Atomkern, den massetragenden teil im zentrum eines atoms;

The nucleus concentrates most of the atom's mass. Atomic nuclei consist of electrically positive protons and electrically neutral neutrons. This is easy to understand and a high scoring topic. These are held together by the strongest known fundamental force, called the strong force. Rutherford's alpha scattering experiment 2. Atomkern, den massetragenden teil im zentrum eines atoms; These are held together by the strongest known fundamental force, called the strong force. Binding energy and binding energy per nucleon 9. The nucleus is a collection of particles called protons, which are positively charged, and neutrons, which are electrically neutral.. The binding energy of a nucleus is the energy holding a nucleus together.

Kern (archäologie), feuersteinblock, von dem steinzeitmenschen stücke zur herstellung von werkzeugen abschlugen fruchtkern, das samentragende harte innere einer kern.. Binding energy curve and inferences 10.nuclear forces. In 1911, ernest rutherford discovered that at the core of every atom is a nucleus. 15.01.2013 · the atomic nucleus is the small central part of the atom, with a positive electric charge. Radius of the nucleus and nuclear density 7. 19.10.2021 · atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. The primary subatomic particles in the nuclei of atoms are protons and neutrons or nucleons (except hydrogen nuclei that contain only one proton). 27.07.2009 · from the physical science course by derek owens.

The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes. Radius of the nucleus and nuclear density 7. Atomic nuclei consist of electrically positive protons and electrically neutral neutrons. Atoms and nuclei is one of the most important chapters from modern physics while preparing for all competitive exam because it helps you to understand atoms which are the basic unit of matter. Binding energy curve and inferences 10.nuclear forces. The concept of atoms and nuclei and some other chapters of physics are mixed in miscellaneous questions which are asked in various competitive exams. These are held together by the strongest known fundamental force, called the strong force. The binding energy of a nucleus is the energy holding a nucleus together.. 19.10.2021 · atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons.

Mass energy relation and mass defect 8... Atomic number, mass number and atomic mass unit 6. 15.01.2013 · the atomic nucleus is the small central part of the atom, with a positive electric charge. The nucleus is a collection of particles called protons, which are positively charged, and neutrons, which are electrically neutral. Rutherford's alpha scattering experiment 2. Binding energy curve and inferences 10.nuclear forces.. Binding energy curve and inferences 10.nuclear forces.

27.07.2009 · from the physical science course by derek owens.. These are held together by the strongest known fundamental force, called the strong force. 27.07.2009 · from the physical science course by derek owens. The binding energy of a nucleus is the energy holding a nucleus together. This is easy to understand and a high scoring topic. Distance of closest approach (nuclear size) 3.

27.07.2009 · from the physical science course by derek owens. These are held together by the strongest known fundamental force, called the strong force. Radius of the nucleus and nuclear density 7. Distance of closest approach (nuclear size) 3. Mass energy relation and mass defect 8. Nucleus (lateinisch „kern"), auch nukleus, bedeutet allgemein den funktional wesentlichen kern eines objekts oder einer gruppe.im speziellen steht der begriff für: Atomkern, den massetragenden teil im zentrum eines atoms; The nucleus concentrates most of the atom's mass... Radius of the nucleus and nuclear density 7.

Binding energy and binding energy per nucleon 9.. Atomic number, mass number and atomic mass unit 6. Nucleus occupies an extremely small volume inside the atom. In 1911, ernest rutherford discovered that at the core of every atom is a nucleus. The nucleus is a collection of particles called protons, which are positively charged, and neutrons, which are electrically neutral. The binding energy of a nucleus is the energy holding a nucleus together. 27.07.2009 · from the physical science course by derek owens.

Distance of closest approach (nuclear size) 3. 15.01.2013 · the atomic nucleus is the small central part of the atom, with a positive electric charge. Binding energy and binding energy per nucleon 9. Nucleus occupies an extremely small volume inside the atom. 27.07.2009 · from the physical science course by derek owens.. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom!

Distance of closest approach (nuclear size) 3... Nucleus occupies an extremely small volume inside the atom. 27.07.2009 · from the physical science course by derek owens. In 1911, ernest rutherford discovered that at the core of every atom is a nucleus. Rutherford's alpha scattering experiment 2. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! Binding energy and binding energy per nucleon 9... These are held together by the strongest known fundamental force, called the strong force.

06.07.2021 · atomic nuclei consist of electrically positive protons and electrically neutral neutrons. 15.01.2013 · the atomic nucleus is the small central part of the atom, with a positive electric charge. These are held together by the strongest known fundamental force, called the strong force. Rutherford's alpha scattering experiment 2.

15.01.2013 · the atomic nucleus is the small central part of the atom, with a positive electric charge. These are held together by the strongest known fundamental force, called the strong force. Mass energy relation and mass defect 8. Distance of closest approach (nuclear size) 3. Atoms and nuclei is one of the most important chapters from modern physics while preparing for all competitive exam because it helps you to understand atoms which are the basic unit of matter. The binding energy of a nucleus is the energy holding a nucleus together. The nucleus concentrates most of the atom's mass. Nucleus (lateinisch „kern"), auch nukleus, bedeutet allgemein den funktional wesentlichen kern eines objekts oder einer gruppe.im speziellen steht der begriff für: 06.07.2021 · atomic nuclei consist of electrically positive protons and electrically neutral neutrons. In 1911, ernest rutherford discovered that at the core of every atom is a nucleus. The nucleus is a collection of particles called protons, which are positively charged, and neutrons, which are electrically neutral. Mass energy relation and mass defect 8.

Nucleus occupies an extremely small volume inside the atom. Distance of closest approach (nuclear size) 3. Binding energy and binding energy per nucleon 9. The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom. The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom. The concept of atoms and nuclei and some other chapters of physics are mixed in miscellaneous questions which are asked in various competitive exams. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! These are held together by the strongest known fundamental force, called the strong force. The nucleus concentrates most of the atom's mass.. In 1911, ernest rutherford discovered that at the core of every atom is a nucleus.

Binding energy curve and inferences 10.nuclear forces. Nucleus (lateinisch „kern"), auch nukleus, bedeutet allgemein den funktional wesentlichen kern eines objekts oder einer gruppe.im speziellen steht der begriff für: The nucleus is a collection of particles called protons, which are positively charged, and neutrons, which are electrically neutral. Rutherford's alpha scattering experiment 2. Atoms and nuclei is one of the most important chapters from modern physics while preparing for all competitive exam because it helps you to understand atoms which are the basic unit of matter... These are held together by the strongest known fundamental force, called the strong force.

Atomkern, den massetragenden teil im zentrum eines atoms; These are held together by the strongest known fundamental force, called the strong force. The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! 15.01.2013 · the atomic nucleus is the small central part of the atom, with a positive electric charge. Distance of closest approach (nuclear size) 3. In 1911, ernest rutherford discovered that at the core of every atom is a nucleus. The nucleus is a collection of particles called protons, which are positively charged, and neutrons, which are electrically neutral. Atoms and nuclei is one of the most important chapters from modern physics while preparing for all competitive exam because it helps you to understand atoms which are the basic unit of matter. Nucleus (lateinisch „kern"), auch nukleus, bedeutet allgemein den funktional wesentlichen kern eines objekts oder einer gruppe.im speziellen steht der begriff für: The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes.. The concept of atoms and nuclei and some other chapters of physics are mixed in miscellaneous questions which are asked in various competitive exams.

19.10.2021 · atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. Atoms and nuclei is one of the most important chapters from modern physics while preparing for all competitive exam because it helps you to understand atoms which are the basic unit of matter. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! Atomic number, mass number and atomic mass unit 6. These are held together by the strongest known fundamental force, called the strong force. These are held together by the strongest known fundamental force, called the strong force.. Atomkern, den massetragenden teil im zentrum eines atoms;

These are held together by the strongest known fundamental force, called the strong force. The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes. Atomkern, den massetragenden teil im zentrum eines atoms; 15.01.2013 · the atomic nucleus is the small central part of the atom, with a positive electric charge. The nucleus is a collection of particles called protons, which are positively charged, and neutrons, which are electrically neutral. These are held together by the strongest known fundamental force, called the strong force. Binding energy and binding energy per nucleon 9. In 1911, ernest rutherford discovered that at the core of every atom is a nucleus. The primary subatomic particles in the nuclei of atoms are protons and neutrons or nucleons (except hydrogen nuclei that contain only one proton).
The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom. The concept of atoms and nuclei and some other chapters of physics are mixed in miscellaneous questions which are asked in various competitive exams. Mass energy relation and mass defect 8. Nucleus (lateinisch „kern"), auch nukleus, bedeutet allgemein den funktional wesentlichen kern eines objekts oder einer gruppe.im speziellen steht der begriff für: In 1911, ernest rutherford discovered that at the core of every atom is a nucleus. The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes. This is easy to understand and a high scoring topic.. The nucleus is a collection of particles called protons, which are positively charged, and neutrons, which are electrically neutral.

19.10.2021 · atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. Mass energy relation and mass defect 8. Nucleus occupies an extremely small volume inside the atom. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom!. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom!

The binding energy of a nucleus is the energy holding a nucleus together... Atoms and nuclei is one of the most important chapters from modern physics while preparing for all competitive exam because it helps you to understand atoms which are the basic unit of matter. The nucleus is a collection of particles called protons, which are positively charged, and neutrons, which are electrically neutral. Atomkern, den massetragenden teil im zentrum eines atoms; These are held together by the strongest known fundamental force, called the strong force. Nucleus occupies an extremely small volume inside the atom. Radius of the nucleus and nuclear density 7. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! Atomic number, mass number and atomic mass unit 6.. The nucleus is a collection of particles called protons, which are positively charged, and neutrons, which are electrically neutral.

These are held together by the strongest known fundamental force, called the strong force. Distance of closest approach (nuclear size) 3. The nucleus is a collection of particles called protons, which are positively charged, and neutrons, which are electrically neutral. Nucleus (lateinisch „kern"), auch nukleus, bedeutet allgemein den funktional wesentlichen kern eines objekts oder einer gruppe.im speziellen steht der begriff für: In 1911, ernest rutherford discovered that at the core of every atom is a nucleus. Rutherford's alpha scattering experiment 2. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! Mass energy relation and mass defect 8. 19.10.2021 · atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom. Atoms and nuclei is one of the most important chapters from modern physics while preparing for all competitive exam because it helps you to understand atoms which are the basic unit of matter.. Mass energy relation and mass defect 8.

19.10.2021 · atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes. Atoms and nuclei is one of the most important chapters from modern physics while preparing for all competitive exam because it helps you to understand atoms which are the basic unit of matter.. The primary subatomic particles in the nuclei of atoms are protons and neutrons or nucleons (except hydrogen nuclei that contain only one proton).

Atomic number, mass number and atomic mass unit 6. Nucleus occupies an extremely small volume inside the atom. The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom. Atoms and nuclei is one of the most important chapters from modern physics while preparing for all competitive exam because it helps you to understand atoms which are the basic unit of matter. Atomkern, den massetragenden teil im zentrum eines atoms; The concept of atoms and nuclei and some other chapters of physics are mixed in miscellaneous questions which are asked in various competitive exams. Atomic number, mass number and atomic mass unit 6... In 1911, ernest rutherford discovered that at the core of every atom is a nucleus.

In 1911, ernest rutherford discovered that at the core of every atom is a nucleus. Binding energy and binding energy per nucleon 9. Atomkern, den massetragenden teil im zentrum eines atoms; The nucleus concentrates most of the atom's mass. Distance of closest approach (nuclear size) 3. Atomic number, mass number and atomic mass unit 6. Nucleus occupies an extremely small volume inside the atom.

The primary subatomic particles in the nuclei of atoms are protons and neutrons or nucleons (except hydrogen nuclei that contain only one proton).. The nucleus concentrates most of the atom's mass. The binding energy of a nucleus is the energy holding a nucleus together. Mass energy relation and mass defect 8.
Nucleus occupies an extremely small volume inside the atom. .. 06.07.2021 · atomic nuclei consist of electrically positive protons and electrically neutral neutrons.
