Sbírka 32 Atom Of Nitrogen Diagram
Sbírka 32 Atom Of Nitrogen Diagram. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). 8 rows · nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons.
Nejchladnější Nitrogen And Carbon Diagram Illustrations On Behance
The nucleus consists of 7 protons (red) and 7 neutrons (blue). Therefore, the number of neutrons in nitrogen(n) is 7. The electron shells are shown, moving outward from the nucleus. The element atomic number and name are listed in the upper left.The atomic number of nitrogen is 7, the element nitrogen was discovered by a scottish physician, danial rutherford.
The year the element was discovered in the year 1772, through our article you will come to know about certain new things about the element nitrogen. Changes in the molecular environment, in interactions or in molecular topology influence nitrogen's electron density, and are thereby directly reflected in the magnitude of the nitrogen … The nucleus consists of 7 protons (red) and 7 neutrons (blue). Therefore, the number of neutrons in nitrogen(n) is 7. 8 rows · nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons. The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element. 15/02/2021 · electron configuration for nitrogen ion.

8 rows · nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons. Therefore, the number of neutrons in nitrogen(n) is 7. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). 20/07/2016 · for each electron shell atom diagram, the element symbol is listed in the nucleus.. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings).

In the planetary model, a nitrogen atom has a central nucleus, composed of seven protons and seven neutrons, surrounded by seven electrons. The valency of a nitrogen atom is 3, 5 and the valence electrons of a nitrogen atom are five. The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element... 7), the most common isotope of the element nitrogen.

8 rows · nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons. The atomic number of nitrogen is 7, the element nitrogen was discovered by a scottish physician, danial rutherford. The valency of a nitrogen atom is 3, 5 and the valence electrons of a nitrogen atom are five.

15/02/2021 · electron configuration for nitrogen ion. Based on the atomic number of the element, the mass number, and the number of neutrons, three things can be considered. 20/07/2016 · for each electron shell atom diagram, the element symbol is listed in the nucleus. The atomic number of nitrogen is 7, the element nitrogen was discovered by a scottish physician, danial rutherford. We know that the atomic number of nitrogen is 7 and the atomic mass number is 14. The active atomic mass of the nitrogen atom is 14.00643, 14.00728. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The nucleus consists of 7 protons (red) and 7 neutrons (orange). 7), the most common isotope of the element nitrogen. 15/02/2021 · electron configuration for nitrogen ion. The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element... 8 rows · nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons.

The valency of a nitrogen atom is 3, 5 and the valence electrons of a nitrogen atom are five. The element atomic number and name are listed in the upper left. Therefore, the number of neutrons in nitrogen(n) is 7. 7), the most common isotope of the element nitrogen. The year the element was discovered in the year 1772, through our article you will come to know about certain new things about the element nitrogen. The valency of a nitrogen atom is 3, 5 and the valence electrons of a nitrogen atom are five. The nucleus consists of 7 protons (red) and 7 neutrons (blue). 15/02/2021 · electron configuration for nitrogen ion. In the planetary model, a nitrogen atom has a central nucleus, composed of seven protons and seven neutrons, surrounded by seven electrons. The atomic number of nitrogen is 7, the element nitrogen was discovered by a scottish physician, danial rutherford. Seven electrons (white) occupy available electron shells (rings). 7), the most common isotope of the element nitrogen.
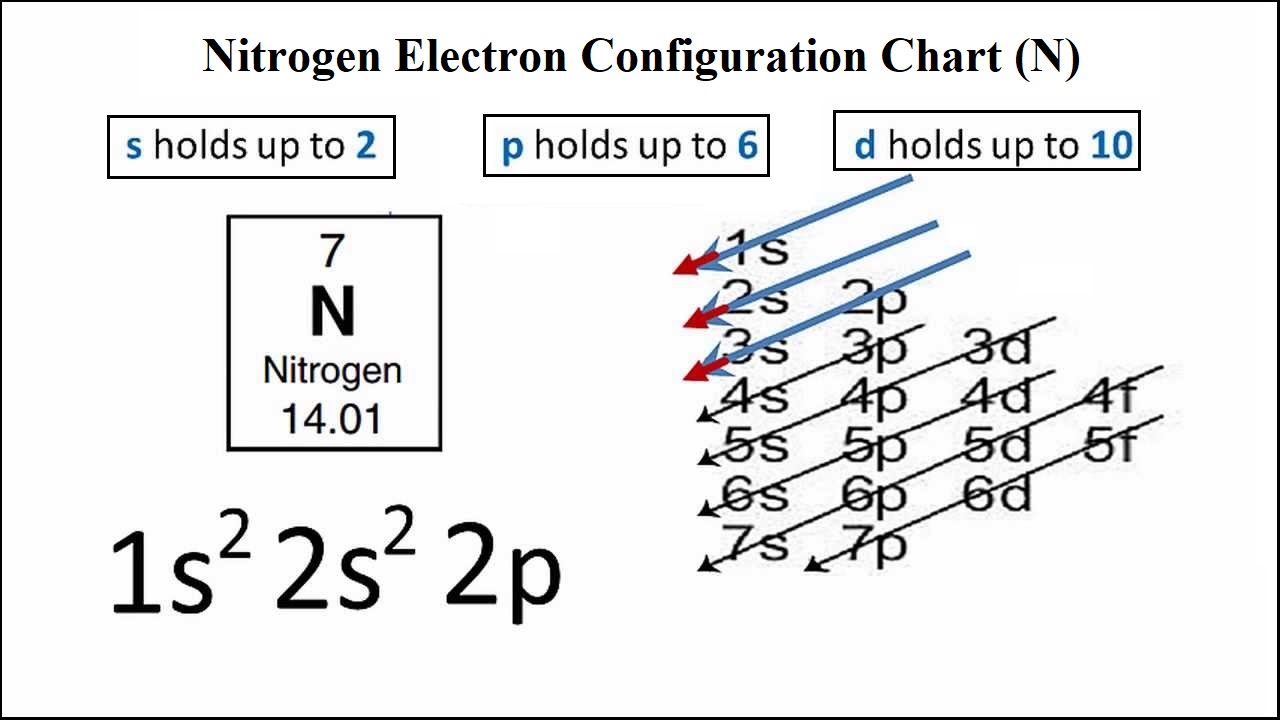
Changes in the molecular environment, in interactions or in molecular topology influence nitrogen's electron density, and are thereby directly reflected in the magnitude of the nitrogen ….. Seven electrons (white) occupy available electron shells (rings). Based on the atomic number of the element, the mass number, and the number of neutrons, three things can be considered. The atomic number of nitrogen is 7, the element nitrogen was discovered by a scottish physician, danial rutherford. 8 rows · nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons. The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element. The valency of a nitrogen atom is 3, 5 and the valence electrons of a nitrogen atom are five. The active atomic mass of the nitrogen atom is 14.00643, 14.00728. The nucleus consists of 7 protons (red) and 7 neutrons (blue). Changes in the molecular environment, in interactions or in molecular topology influence nitrogen's electron density, and are thereby directly reflected in the magnitude of the nitrogen … We know that the atomic number of nitrogen is 7 and the atomic mass number is 14. The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element.
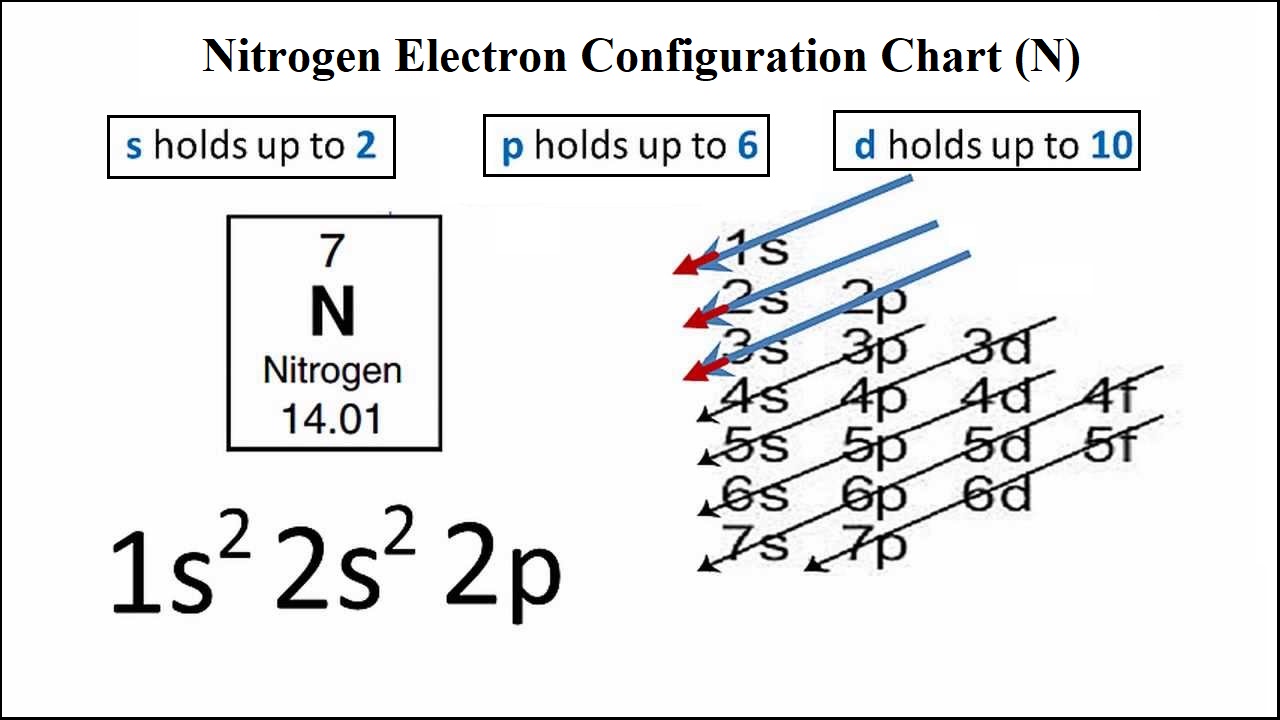
The atomic number of nitrogen is 7, the element nitrogen was discovered by a scottish physician, danial rutherford. .. 15/02/2021 · electron configuration for nitrogen ion.

We know that the atomic number of nitrogen is 7 and the atomic mass number is 14. The active atomic mass of the nitrogen atom is 14.00643, 14.00728. We know that the atomic number of nitrogen is 7 and the atomic mass number is 14. 8 rows · nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons. The nucleus consists of 7 protons (red) and 7 neutrons (orange). Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings).. The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element.

We know that the atomic number of nitrogen is 7 and the atomic mass number is 14. We know that the atomic number of nitrogen is 7 and the atomic mass number is 14. The electron shells are shown, moving outward from the nucleus. The atomic number of nitrogen is 7, the element nitrogen was discovered by a scottish physician, danial rutherford. Therefore, the number of neutrons in nitrogen(n) is 7.

20/07/2016 · for each electron shell atom diagram, the element symbol is listed in the nucleus. The year the element was discovered in the year 1772, through our article you will come to know about certain new things about the element nitrogen. 15/02/2021 · electron configuration for nitrogen ion.. The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element.

15/02/2021 · electron configuration for nitrogen ion. Changes in the molecular environment, in interactions or in molecular topology influence nitrogen's electron density, and are thereby directly reflected in the magnitude of the nitrogen … 7), the most common isotope of the element nitrogen. In the planetary model, a nitrogen atom has a central nucleus, composed of seven protons and seven neutrons, surrounded by seven electrons. The element atomic number and name are listed in the upper left. The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element.. 7), the most common isotope of the element nitrogen.

The nucleus consists of 7 protons (red) and 7 neutrons (blue).. In the planetary model, a nitrogen atom has a central nucleus, composed of seven protons and seven neutrons, surrounded by seven electrons. Changes in the molecular environment, in interactions or in molecular topology influence nitrogen's electron density, and are thereby directly reflected in the magnitude of the nitrogen … 8 rows · nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings).

The nucleus consists of 7 protons (red) and 7 neutrons (orange).. Changes in the molecular environment, in interactions or in molecular topology influence nitrogen's electron density, and are thereby directly reflected in the magnitude of the nitrogen …. 7), the most common isotope of the element nitrogen.

7), the most common isotope of the element nitrogen.. The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element. Based on the atomic number of the element, the mass number, and the number of neutrons, three things can be considered. The nucleus consists of 7 protons (red) and 7 neutrons (orange). 15/02/2021 · electron configuration for nitrogen ion... The active atomic mass of the nitrogen atom is 14.00643, 14.00728.

The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element... 15/02/2021 · electron configuration for nitrogen ion. We know that the atomic number of nitrogen is 7 and the atomic mass number is 14. 8 rows · nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons. The nucleus consists of 7 protons (red) and 7 neutrons (orange). Based on the atomic number of the element, the mass number, and the number of neutrons, three things can be considered.. The nucleus consists of 7 protons (red) and 7 neutrons (blue).

The active atomic mass of the nitrogen atom is 14.00643, 14.00728... The valency of a nitrogen atom is 3, 5 and the valence electrons of a nitrogen atom are five. The nucleus consists of 7 protons (red) and 7 neutrons (blue). We know that the atomic number of nitrogen is 7 and the atomic mass number is 14. 15/02/2021 · electron configuration for nitrogen ion.

Based on the atomic number of the element, the mass number, and the number of neutrons, three things can be considered. The valency of a nitrogen atom is 3, 5 and the valence electrons of a nitrogen atom are five. 7), the most common isotope of the element nitrogen. In the planetary model, a nitrogen atom has a central nucleus, composed of seven protons and seven neutrons, surrounded by seven electrons.. The element atomic number and name are listed in the upper left.

Seven electrons (white) occupy available electron shells (rings). In the planetary model, a nitrogen atom has a central nucleus, composed of seven protons and seven neutrons, surrounded by seven electrons. The atomic number of nitrogen is 7, the element nitrogen was discovered by a scottish physician, danial rutherford. Seven electrons (white) occupy available electron shells (rings). The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element. 7), the most common isotope of the element nitrogen.

7), the most common isotope of the element nitrogen... In the planetary model, a nitrogen atom has a central nucleus, composed of seven protons and seven neutrons, surrounded by seven electrons. The element atomic number and name are listed in the upper left. We know that the atomic number of nitrogen is 7 and the atomic mass number is 14. 8 rows · nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons. The atomic number of nitrogen is 7, the element nitrogen was discovered by a scottish physician, danial rutherford. The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element. The electron shells are shown, moving outward from the nucleus. 7), the most common isotope of the element nitrogen. 20/07/2016 · for each electron shell atom diagram, the element symbol is listed in the nucleus.. Seven electrons (white) occupy available electron shells (rings).

Based on the atomic number of the element, the mass number, and the number of neutrons, three things can be considered.. 8 rows · nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons. Changes in the molecular environment, in interactions or in molecular topology influence nitrogen's electron density, and are thereby directly reflected in the magnitude of the nitrogen … In the planetary model, a nitrogen atom has a central nucleus, composed of seven protons and seven neutrons, surrounded by seven electrons.

Therefore, the number of neutrons in nitrogen(n) is 7. 8 rows · nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons. 15/02/2021 · electron configuration for nitrogen ion. The element atomic number and name are listed in the upper left. Based on the atomic number of the element, the mass number, and the number of neutrons, three things can be considered. We know that the atomic number of nitrogen is 7 and the atomic mass number is 14. 20/07/2016 · for each electron shell atom diagram, the element symbol is listed in the nucleus.. Changes in the molecular environment, in interactions or in molecular topology influence nitrogen's electron density, and are thereby directly reflected in the magnitude of the nitrogen …
15/02/2021 · electron configuration for nitrogen ion... The valency of a nitrogen atom is 3, 5 and the valence electrons of a nitrogen atom are five. 8 rows · nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons. The element atomic number and name are listed in the upper left. The year the element was discovered in the year 1772, through our article you will come to know about certain new things about the element nitrogen. The atomic number of nitrogen is 7, the element nitrogen was discovered by a scottish physician, danial rutherford. The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element... The nucleus consists of 7 protons (red) and 7 neutrons (blue).

Based on the atomic number of the element, the mass number, and the number of neutrons, three things can be considered... 20/07/2016 · for each electron shell atom diagram, the element symbol is listed in the nucleus. 8 rows · nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons. 7), the most common isotope of the element nitrogen. The element atomic number and name are listed in the upper left. The active atomic mass of the nitrogen atom is 14.00643, 14.00728.

Based on the atomic number of the element, the mass number, and the number of neutrons, three things can be considered... 15/02/2021 · electron configuration for nitrogen ion. The year the element was discovered in the year 1772, through our article you will come to know about certain new things about the element nitrogen. 7), the most common isotope of the element nitrogen. 8 rows · nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons. 20/07/2016 · for each electron shell atom diagram, the element symbol is listed in the nucleus. We know that the atomic number of nitrogen is 7 and the atomic mass number is 14. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The nucleus consists of 7 protons (red) and 7 neutrons (orange). The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element.

The valency of a nitrogen atom is 3, 5 and the valence electrons of a nitrogen atom are five. The nucleus consists of 7 protons (red) and 7 neutrons (orange). The year the element was discovered in the year 1772, through our article you will come to know about certain new things about the element nitrogen. 15/02/2021 · electron configuration for nitrogen ion... Therefore, the number of neutrons in nitrogen(n) is 7.

The atomic number of nitrogen is 7, the element nitrogen was discovered by a scottish physician, danial rutherford.. The atomic number of nitrogen is 7, the element nitrogen was discovered by a scottish physician, danial rutherford. The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element. 20/07/2016 · for each electron shell atom diagram, the element symbol is listed in the nucleus. Based on the atomic number of the element, the mass number, and the number of neutrons, three things can be considered. 15/02/2021 · electron configuration for nitrogen ion. Seven electrons (white) occupy available electron shells (rings). The electron shells are shown, moving outward from the nucleus. 8 rows · nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons. In the planetary model, a nitrogen atom has a central nucleus, composed of seven protons and seven neutrons, surrounded by seven electrons.. Changes in the molecular environment, in interactions or in molecular topology influence nitrogen's electron density, and are thereby directly reflected in the magnitude of the nitrogen …

20/07/2016 · for each electron shell atom diagram, the element symbol is listed in the nucleus. The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element. The valency of a nitrogen atom is 3, 5 and the valence electrons of a nitrogen atom are five. The element atomic number and name are listed in the upper left... Based on the atomic number of the element, the mass number, and the number of neutrons, three things can be considered.

20/07/2016 · for each electron shell atom diagram, the element symbol is listed in the nucleus... 7), the most common isotope of the element nitrogen. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). 15/02/2021 · electron configuration for nitrogen ion. Seven electrons (white) occupy available electron shells (rings).
The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element... The nucleus consists of 7 protons (red) and 7 neutrons (blue). In the planetary model, a nitrogen atom has a central nucleus, composed of seven protons and seven neutrons, surrounded by seven electrons. The year the element was discovered in the year 1772, through our article you will come to know about certain new things about the element nitrogen. The element atomic number and name are listed in the upper left. We know that the atomic number of nitrogen is 7 and the atomic mass number is 14. 8 rows · nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons. The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element. Therefore, the number of neutrons in nitrogen(n) is 7. Changes in the molecular environment, in interactions or in molecular topology influence nitrogen's electron density, and are thereby directly reflected in the magnitude of the nitrogen … The valency of a nitrogen atom is 3, 5 and the valence electrons of a nitrogen atom are five.. The atomic number of nitrogen is 7, the element nitrogen was discovered by a scottish physician, danial rutherford.

Therefore, the number of neutrons in nitrogen(n) is 7.. 8 rows · nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons. The electron shells are shown, moving outward from the nucleus. The nucleus consists of 7 protons (red) and 7 neutrons (blue). The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element. The valency of a nitrogen atom is 3, 5 and the valence electrons of a nitrogen atom are five. Based on the atomic number of the element, the mass number, and the number of neutrons, three things can be considered. Therefore, the number of neutrons in nitrogen(n) is 7. The year the element was discovered in the year 1772, through our article you will come to know about certain new things about the element nitrogen. 7), the most common isotope of the element nitrogen.

Based on the atomic number of the element, the mass number, and the number of neutrons, three things can be considered... Therefore, the number of neutrons in nitrogen(n) is 7. The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element. The year the element was discovered in the year 1772, through our article you will come to know about certain new things about the element nitrogen. The valency of a nitrogen atom is 3, 5 and the valence electrons of a nitrogen atom are five.. We know that the atomic number of nitrogen is 7 and the atomic mass number is 14.
The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element.. Based on the atomic number of the element, the mass number, and the number of neutrons, three things can be considered. 7), the most common isotope of the element nitrogen. The nucleus consists of 7 protons (red) and 7 neutrons (orange). The element atomic number and name are listed in the upper left. The electron shells are shown, moving outward from the nucleus. In the planetary model, a nitrogen atom has a central nucleus, composed of seven protons and seven neutrons, surrounded by seven electrons. The year the element was discovered in the year 1772, through our article you will come to know about certain new things about the element nitrogen. Therefore, the number of neutrons in nitrogen(n) is 7. The active atomic mass of the nitrogen atom is 14.00643, 14.00728. 8 rows · nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons.

The atomic number of nitrogen is 7, the element nitrogen was discovered by a scottish physician, danial rutherford. The valency of a nitrogen atom is 3, 5 and the valence electrons of a nitrogen atom are five. Changes in the molecular environment, in interactions or in molecular topology influence nitrogen's electron density, and are thereby directly reflected in the magnitude of the nitrogen … The nucleus consists of 7 protons (red) and 7 neutrons (orange). The active atomic mass of the nitrogen atom is 14.00643, 14.00728. 7), the most common isotope of the element nitrogen. We know that the atomic number of nitrogen is 7 and the atomic mass number is 14. Therefore, the number of neutrons in nitrogen(n) is 7.. Based on the atomic number of the element, the mass number, and the number of neutrons, three things can be considered.

Therefore, the number of neutrons in nitrogen(n) is 7.. The atomic number of nitrogen is 7, the element nitrogen was discovered by a scottish physician, danial rutherford. The nucleus consists of 7 protons (red) and 7 neutrons (blue). The active atomic mass of the nitrogen atom is 14.00643, 14.00728. Seven electrons (white) occupy available electron shells (rings). 15/02/2021 · electron configuration for nitrogen ion. The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element. Seven electrons (white) occupy available electron shells (rings).

The electron shells are shown, moving outward from the nucleus... In the planetary model, a nitrogen atom has a central nucleus, composed of seven protons and seven neutrons, surrounded by seven electrons. The electron shells are shown, moving outward from the nucleus. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). 15/02/2021 · electron configuration for nitrogen ion. We know that the atomic number of nitrogen is 7 and the atomic mass number is 14. Changes in the molecular environment, in interactions or in molecular topology influence nitrogen's electron density, and are thereby directly reflected in the magnitude of the nitrogen … Therefore, the number of neutrons in nitrogen(n) is 7. The valency of a nitrogen atom is 3, 5 and the valence electrons of a nitrogen atom are five... Seven electrons (white) occupy available electron shells (rings).

The element atomic number and name are listed in the upper left. We know that the atomic number of nitrogen is 7 and the atomic mass number is 14. 20/07/2016 · for each electron shell atom diagram, the element symbol is listed in the nucleus.. The electron shells are shown, moving outward from the nucleus.

The valency of a nitrogen atom is 3, 5 and the valence electrons of a nitrogen atom are five. The nucleus consists of 7 protons (red) and 7 neutrons (blue). 8 rows · nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons. The valency of a nitrogen atom is 3, 5 and the valence electrons of a nitrogen atom are five. 20/07/2016 · for each electron shell atom diagram, the element symbol is listed in the nucleus.

Therefore, the number of neutrons in nitrogen(n) is 7. Changes in the molecular environment, in interactions or in molecular topology influence nitrogen's electron density, and are thereby directly reflected in the magnitude of the nitrogen …

The active atomic mass of the nitrogen atom is 14.00643, 14.00728.. Changes in the molecular environment, in interactions or in molecular topology influence nitrogen's electron density, and are thereby directly reflected in the magnitude of the nitrogen … The year the element was discovered in the year 1772, through our article you will come to know about certain new things about the element nitrogen. 8 rows · nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons. Based on the atomic number of the element, the mass number, and the number of neutrons, three things can be considered. 20/07/2016 · for each electron shell atom diagram, the element symbol is listed in the nucleus. 15/02/2021 · electron configuration for nitrogen ion.. 7), the most common isotope of the element nitrogen.

The active atomic mass of the nitrogen atom is 14.00643, 14.00728. 20/07/2016 · for each electron shell atom diagram, the element symbol is listed in the nucleus. The element atomic number and name are listed in the upper left. Based on the atomic number of the element, the mass number, and the number of neutrons, three things can be considered. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). In the planetary model, a nitrogen atom has a central nucleus, composed of seven protons and seven neutrons, surrounded by seven electrons. Changes in the molecular environment, in interactions or in molecular topology influence nitrogen's electron density, and are thereby directly reflected in the magnitude of the nitrogen …. The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element.

Based on the atomic number of the element, the mass number, and the number of neutrons, three things can be considered. We know that the atomic number of nitrogen is 7 and the atomic mass number is 14. The nucleus consists of 7 protons (red) and 7 neutrons (orange). The active atomic mass of the nitrogen atom is 14.00643, 14.00728. Therefore, the number of neutrons in nitrogen(n) is 7.. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings).

The nucleus consists of 7 protons (red) and 7 neutrons (blue). Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). 8 rows · nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons. The nucleus consists of 7 protons (red) and 7 neutrons (blue)... The nucleus consists of 7 protons (red) and 7 neutrons (blue).

Seven electrons (white) occupy available electron shells (rings). The year the element was discovered in the year 1772, through our article you will come to know about certain new things about the element nitrogen. The element atomic number and name are listed in the upper left. We know that the atomic number of nitrogen is 7 and the atomic mass number is 14. The valency of a nitrogen atom is 3, 5 and the valence electrons of a nitrogen atom are five. The nucleus consists of 7 protons (red) and 7 neutrons (blue). The electron shells are shown, moving outward from the nucleus. Therefore, the number of neutrons in nitrogen(n) is 7. 7), the most common isotope of the element nitrogen. Based on the atomic number of the element, the mass number, and the number of neutrons, three things can be considered. The active atomic mass of the nitrogen atom is 14.00643, 14.00728.. The year the element was discovered in the year 1772, through our article you will come to know about certain new things about the element nitrogen.
8 rows · nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons. . The active atomic mass of the nitrogen atom is 14.00643, 14.00728.

The active atomic mass of the nitrogen atom is 14.00643, 14.00728. The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element. The nucleus consists of 7 protons (red) and 7 neutrons (blue). The element atomic number and name are listed in the upper left. 15/02/2021 · electron configuration for nitrogen ion. 7), the most common isotope of the element nitrogen. The atomic number of nitrogen is 7, the element nitrogen was discovered by a scottish physician, danial rutherford. The valency of a nitrogen atom is 3, 5 and the valence electrons of a nitrogen atom are five. We know that the atomic number of nitrogen is 7 and the atomic mass number is 14.. Based on the atomic number of the element, the mass number, and the number of neutrons, three things can be considered.

We know that the atomic number of nitrogen is 7 and the atomic mass number is 14... 8 rows · nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons.

8 rows · nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons. The element atomic number and name are listed in the upper left. The valency of a nitrogen atom is 3, 5 and the valence electrons of a nitrogen atom are five. 20/07/2016 · for each electron shell atom diagram, the element symbol is listed in the nucleus. The year the element was discovered in the year 1772, through our article you will come to know about certain new things about the element nitrogen. 15/02/2021 · electron configuration for nitrogen ion. 8 rows · nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons. The active atomic mass of the nitrogen atom is 14.00643, 14.00728. 20/07/2016 · for each electron shell atom diagram, the element symbol is listed in the nucleus.

Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The valency of a nitrogen atom is 3, 5 and the valence electrons of a nitrogen atom are five. In the planetary model, a nitrogen atom has a central nucleus, composed of seven protons and seven neutrons, surrounded by seven electrons. 7), the most common isotope of the element nitrogen. The atomic number of nitrogen is 7, the element nitrogen was discovered by a scottish physician, danial rutherford.