Nápady 108+ Atom Economy Definition
Nápady 108+ Atom Economy Definition. Percentage yield and atom economy show how much desired product is obtained compared to amounts of starting materials. Atom economy was designed to overcome the limitations of the traditional concept of "yield," the amount of final products, which was… Other articles where atom economy is discussed: The greater the % atom economy of a reaction, the more 'efficient' or 'economic' it is likely to be, though this is a gross simplification when complex and costly chemical synthesis are looked at. Gas calculations show volumes of gas used and obtained in chemical reactions.
Prezentováno Savvy Chemist Gcse Ocr Gateway Chemistry C5 1i J Atom Economy
Percentage yield and atom economy show how much desired product is obtained compared to amounts of starting materials. Of these principles, "atom economy," originally suggested by american chemist barry trost in 1973, became a central concept among green chemistry researchers. Other articles where atom economy is discussed:In atom economy calculations you can say reactants or products because of the law of conservation of mass.
In an ideal chemical process, the amount of starting materials or reactants equals the amount of all products generated (see stoichiometry) and no atom is wasted.recent developments like high raw material (such as petrochemicals) costs and. Other articles where atom economy is discussed: The atom economy ( atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products*. Atom economy (atom efficiency) describes the conversion efficiency of a chemical process in terms of all atoms involved (desired products produced). Gas calculations show volumes of gas used and obtained in chemical reactions. The greater the % atom economy of a reaction, the more 'efficient' or 'economic' it is likely to be, though this is a gross simplification when complex and costly chemical synthesis are looked at.

In atom economy calculations you can say reactants or products because of the law of conservation of mass. Of these principles, "atom economy," originally suggested by american chemist barry trost in 1973, became a central concept among green chemistry researchers. The greater the value of the % atom economy, the less the amount of waste product produced. Atom economy was designed to overcome the limitations of the traditional concept of "yield," the amount of final products, which was… In atom economy calculations you can say reactants or products because of the law of conservation of mass. Percentage yield and atom economy show how much desired product is obtained compared to amounts of starting materials. Other articles where atom economy is discussed: Atom economy (atom efficiency) describes the conversion efficiency of a chemical process in terms of all atoms involved (desired products produced). The greater the % atom economy of a reaction, the more 'efficient' or 'economic' it is likely to be, though this is a gross simplification when complex and costly chemical synthesis are looked at. Gas calculations show volumes of gas used and obtained in chemical reactions. Other articles where atom economy is discussed:

The atom economy ( atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products*... Atom economy (atom efficiency) describes the conversion efficiency of a chemical process in terms of all atoms involved (desired products produced). Other articles where atom economy is discussed:. In atom economy calculations you can say reactants or products because of the law of conservation of mass.

Other articles where atom economy is discussed:.. The greater the value of the % atom economy, the less the amount of waste product produced. Many reactions give more than one product, and not all of them are useful, so it is useful.

Other articles where atom economy is discussed:. Atom economy (atom efficiency) describes the conversion efficiency of a chemical process in terms of all atoms involved (desired products produced).. Gas calculations show volumes of gas used and obtained in chemical reactions.

Percentage yield and atom economy show how much desired product is obtained compared to amounts of starting materials... . The atom economy ( atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products*.

Of these principles, "atom economy," originally suggested by american chemist barry trost in 1973, became a central concept among green chemistry researchers.. Atom economy (atom efficiency) describes the conversion efficiency of a chemical process in terms of all atoms involved (desired products produced). Atom economy was designed to overcome the limitations of the traditional concept of "yield," the amount of final products, which was… Other articles where atom economy is discussed: In an ideal chemical process, the amount of starting materials or reactants equals the amount of all products generated (see stoichiometry) and no atom is wasted.recent developments like high raw material (such as petrochemicals) costs and. Gas calculations show volumes of gas used and obtained in chemical reactions. Percentage yield and atom economy show how much desired product is obtained compared to amounts of starting materials. Percentage yield and atom economy show how much desired product is obtained compared to amounts of starting materials.

Atom economy was designed to overcome the limitations of the traditional concept of "yield," the amount of final products, which was…. The greater the % atom economy of a reaction, the more 'efficient' or 'economic' it is likely to be, though this is a gross simplification when complex and costly chemical synthesis are looked at. In atom economy calculations you can say reactants or products because of the law of conservation of mass. Of these principles, "atom economy," originally suggested by american chemist barry trost in 1973, became a central concept among green chemistry researchers. Percentage yield and atom economy show how much desired product is obtained compared to amounts of starting materials.

In an ideal chemical process, the amount of starting materials or reactants equals the amount of all products generated (see stoichiometry) and no atom is wasted.recent developments like high raw material (such as petrochemicals) costs and. In atom economy calculations you can say reactants or products because of the law of conservation of mass. Of these principles, "atom economy," originally suggested by american chemist barry trost in 1973, became a central concept among green chemistry researchers. The greater the % atom economy of a reaction, the more 'efficient' or 'economic' it is likely to be, though this is a gross simplification when complex and costly chemical synthesis are looked at. The greater the value of the % atom economy, the less the amount of waste product produced.

The greater the % atom economy of a reaction, the more 'efficient' or 'economic' it is likely to be, though this is a gross simplification when complex and costly chemical synthesis are looked at. The greater the value of the % atom economy, the less the amount of waste product produced. The greater the % atom economy of a reaction, the more 'efficient' or 'economic' it is likely to be, though this is a gross simplification when complex and costly chemical synthesis are looked at. Percentage yield and atom economy show how much desired product is obtained compared to amounts of starting materials. Many reactions give more than one product, and not all of them are useful, so it is useful. Of these principles, "atom economy," originally suggested by american chemist barry trost in 1973, became a central concept among green chemistry researchers.. Of these principles, "atom economy," originally suggested by american chemist barry trost in 1973, became a central concept among green chemistry researchers.

Other articles where atom economy is discussed: Atom economy was designed to overcome the limitations of the traditional concept of "yield," the amount of final products, which was… The greater the % atom economy of a reaction, the more 'efficient' or 'economic' it is likely to be, though this is a gross simplification when complex and costly chemical synthesis are looked at. Of these principles, "atom economy," originally suggested by american chemist barry trost in 1973, became a central concept among green chemistry researchers. In atom economy calculations you can say reactants or products because of the law of conservation of mass. Gas calculations show volumes of gas used and obtained in chemical reactions. Other articles where atom economy is discussed: Many reactions give more than one product, and not all of them are useful, so it is useful. The greater the value of the % atom economy, the less the amount of waste product produced. Percentage yield and atom economy show how much desired product is obtained compared to amounts of starting materials. In an ideal chemical process, the amount of starting materials or reactants equals the amount of all products generated (see stoichiometry) and no atom is wasted.recent developments like high raw material (such as petrochemicals) costs and... In atom economy calculations you can say reactants or products because of the law of conservation of mass.

The greater the % atom economy of a reaction, the more 'efficient' or 'economic' it is likely to be, though this is a gross simplification when complex and costly chemical synthesis are looked at.. The atom economy ( atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products*. Of these principles, "atom economy," originally suggested by american chemist barry trost in 1973, became a central concept among green chemistry researchers. The greater the % atom economy of a reaction, the more 'efficient' or 'economic' it is likely to be, though this is a gross simplification when complex and costly chemical synthesis are looked at. The greater the value of the % atom economy, the less the amount of waste product produced. The atom economy ( atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products*.

In atom economy calculations you can say reactants or products because of the law of conservation of mass. The atom economy ( atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products*.. Percentage yield and atom economy show how much desired product is obtained compared to amounts of starting materials.

Other articles where atom economy is discussed: Other articles where atom economy is discussed: In an ideal chemical process, the amount of starting materials or reactants equals the amount of all products generated (see stoichiometry) and no atom is wasted.recent developments like high raw material (such as petrochemicals) costs and. The greater the % atom economy of a reaction, the more 'efficient' or 'economic' it is likely to be, though this is a gross simplification when complex and costly chemical synthesis are looked at.. Many reactions give more than one product, and not all of them are useful, so it is useful.

The greater the % atom economy of a reaction, the more 'efficient' or 'economic' it is likely to be, though this is a gross simplification when complex and costly chemical synthesis are looked at. Gas calculations show volumes of gas used and obtained in chemical reactions. Atom economy was designed to overcome the limitations of the traditional concept of "yield," the amount of final products, which was… The atom economy ( atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products*. In an ideal chemical process, the amount of starting materials or reactants equals the amount of all products generated (see stoichiometry) and no atom is wasted.recent developments like high raw material (such as petrochemicals) costs and... Atom economy (atom efficiency) describes the conversion efficiency of a chemical process in terms of all atoms involved (desired products produced).

Gas calculations show volumes of gas used and obtained in chemical reactions.. In atom economy calculations you can say reactants or products because of the law of conservation of mass. The atom economy ( atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products*. In an ideal chemical process, the amount of starting materials or reactants equals the amount of all products generated (see stoichiometry) and no atom is wasted.recent developments like high raw material (such as petrochemicals) costs and.

Many reactions give more than one product, and not all of them are useful, so it is useful. Atom economy was designed to overcome the limitations of the traditional concept of "yield," the amount of final products, which was… Many reactions give more than one product, and not all of them are useful, so it is useful. Percentage yield and atom economy show how much desired product is obtained compared to amounts of starting materials. Of these principles, "atom economy," originally suggested by american chemist barry trost in 1973, became a central concept among green chemistry researchers. Gas calculations show volumes of gas used and obtained in chemical reactions. The greater the % atom economy of a reaction, the more 'efficient' or 'economic' it is likely to be, though this is a gross simplification when complex and costly chemical synthesis are looked at.

In an ideal chemical process, the amount of starting materials or reactants equals the amount of all products generated (see stoichiometry) and no atom is wasted.recent developments like high raw material (such as petrochemicals) costs and. Other articles where atom economy is discussed: The atom economy ( atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products*... Of these principles, "atom economy," originally suggested by american chemist barry trost in 1973, became a central concept among green chemistry researchers.
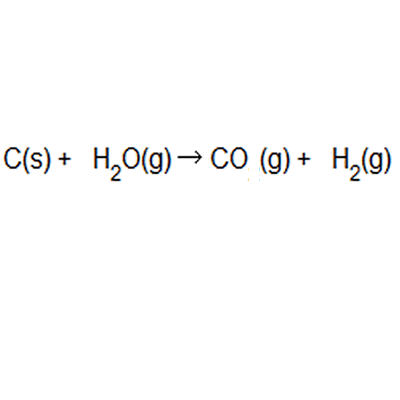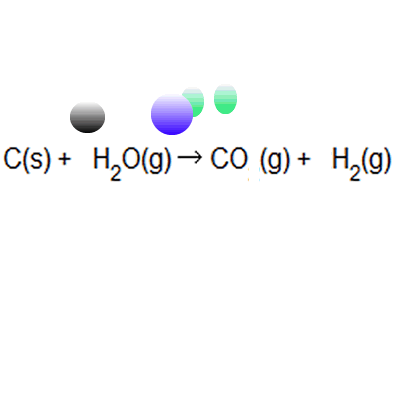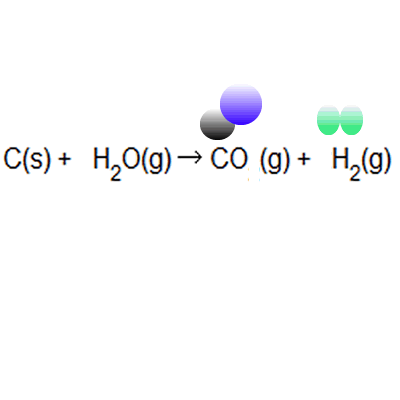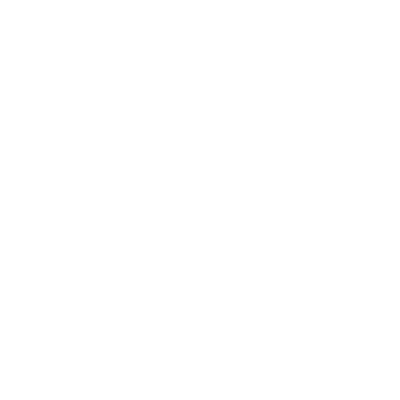
Of these principles, "atom economy," originally suggested by american chemist barry trost in 1973, became a central concept among green chemistry researchers. The greater the value of the % atom economy, the less the amount of waste product produced. In atom economy calculations you can say reactants or products because of the law of conservation of mass. Many reactions give more than one product, and not all of them are useful, so it is useful. The greater the % atom economy of a reaction, the more 'efficient' or 'economic' it is likely to be, though this is a gross simplification when complex and costly chemical synthesis are looked at.. In atom economy calculations you can say reactants or products because of the law of conservation of mass.

In atom economy calculations you can say reactants or products because of the law of conservation of mass. The greater the % atom economy of a reaction, the more 'efficient' or 'economic' it is likely to be, though this is a gross simplification when complex and costly chemical synthesis are looked at. Other articles where atom economy is discussed: Of these principles, "atom economy," originally suggested by american chemist barry trost in 1973, became a central concept among green chemistry researchers. The greater the value of the % atom economy, the less the amount of waste product produced. Atom economy (atom efficiency) describes the conversion efficiency of a chemical process in terms of all atoms involved (desired products produced). The atom economy ( atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products*. Gas calculations show volumes of gas used and obtained in chemical reactions.. Percentage yield and atom economy show how much desired product is obtained compared to amounts of starting materials.

In an ideal chemical process, the amount of starting materials or reactants equals the amount of all products generated (see stoichiometry) and no atom is wasted.recent developments like high raw material (such as petrochemicals) costs and.. Other articles where atom economy is discussed: Many reactions give more than one product, and not all of them are useful, so it is useful. Atom economy (atom efficiency) describes the conversion efficiency of a chemical process in terms of all atoms involved (desired products produced)... Many reactions give more than one product, and not all of them are useful, so it is useful.

Percentage yield and atom economy show how much desired product is obtained compared to amounts of starting materials. The greater the value of the % atom economy, the less the amount of waste product produced. In atom economy calculations you can say reactants or products because of the law of conservation of mass. In an ideal chemical process, the amount of starting materials or reactants equals the amount of all products generated (see stoichiometry) and no atom is wasted.recent developments like high raw material (such as petrochemicals) costs and. Of these principles, "atom economy," originally suggested by american chemist barry trost in 1973, became a central concept among green chemistry researchers... Atom economy was designed to overcome the limitations of the traditional concept of "yield," the amount of final products, which was…

Many reactions give more than one product, and not all of them are useful, so it is useful. Percentage yield and atom economy show how much desired product is obtained compared to amounts of starting materials. The atom economy ( atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products*. The atom economy ( atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products*.

Percentage yield and atom economy show how much desired product is obtained compared to amounts of starting materials.. In atom economy calculations you can say reactants or products because of the law of conservation of mass. The greater the value of the % atom economy, the less the amount of waste product produced. The greater the % atom economy of a reaction, the more 'efficient' or 'economic' it is likely to be, though this is a gross simplification when complex and costly chemical synthesis are looked at. Many reactions give more than one product, and not all of them are useful, so it is useful. Atom economy (atom efficiency) describes the conversion efficiency of a chemical process in terms of all atoms involved (desired products produced). In an ideal chemical process, the amount of starting materials or reactants equals the amount of all products generated (see stoichiometry) and no atom is wasted.recent developments like high raw material (such as petrochemicals) costs and.

Atom economy (atom efficiency) describes the conversion efficiency of a chemical process in terms of all atoms involved (desired products produced).. Atom economy (atom efficiency) describes the conversion efficiency of a chemical process in terms of all atoms involved (desired products produced). Gas calculations show volumes of gas used and obtained in chemical reactions. The greater the value of the % atom economy, the less the amount of waste product produced. The atom economy ( atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products*. In an ideal chemical process, the amount of starting materials or reactants equals the amount of all products generated (see stoichiometry) and no atom is wasted.recent developments like high raw material (such as petrochemicals) costs and. Other articles where atom economy is discussed: The greater the value of the % atom economy, the less the amount of waste product produced.

In atom economy calculations you can say reactants or products because of the law of conservation of mass... Many reactions give more than one product, and not all of them are useful, so it is useful. The greater the % atom economy of a reaction, the more 'efficient' or 'economic' it is likely to be, though this is a gross simplification when complex and costly chemical synthesis are looked at. The atom economy ( atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products*. Percentage yield and atom economy show how much desired product is obtained compared to amounts of starting materials. Other articles where atom economy is discussed: In an ideal chemical process, the amount of starting materials or reactants equals the amount of all products generated (see stoichiometry) and no atom is wasted.recent developments like high raw material (such as petrochemicals) costs and. Atom economy (atom efficiency) describes the conversion efficiency of a chemical process in terms of all atoms involved (desired products produced).

Gas calculations show volumes of gas used and obtained in chemical reactions... The greater the % atom economy of a reaction, the more 'efficient' or 'economic' it is likely to be, though this is a gross simplification when complex and costly chemical synthesis are looked at. Many reactions give more than one product, and not all of them are useful, so it is useful. Percentage yield and atom economy show how much desired product is obtained compared to amounts of starting materials. Gas calculations show volumes of gas used and obtained in chemical reactions. Atom economy was designed to overcome the limitations of the traditional concept of "yield," the amount of final products, which was… In atom economy calculations you can say reactants or products because of the law of conservation of mass. The atom economy ( atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products*. Of these principles, "atom economy," originally suggested by american chemist barry trost in 1973, became a central concept among green chemistry researchers. Of these principles, "atom economy," originally suggested by american chemist barry trost in 1973, became a central concept among green chemistry researchers.

In an ideal chemical process, the amount of starting materials or reactants equals the amount of all products generated (see stoichiometry) and no atom is wasted.recent developments like high raw material (such as petrochemicals) costs and... In atom economy calculations you can say reactants or products because of the law of conservation of mass. Atom economy (atom efficiency) describes the conversion efficiency of a chemical process in terms of all atoms involved (desired products produced). The atom economy ( atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products*. Atom economy was designed to overcome the limitations of the traditional concept of "yield," the amount of final products, which was… In an ideal chemical process, the amount of starting materials or reactants equals the amount of all products generated (see stoichiometry) and no atom is wasted.recent developments like high raw material (such as petrochemicals) costs and. Atom economy (atom efficiency) describes the conversion efficiency of a chemical process in terms of all atoms involved (desired products produced).

The greater the value of the % atom economy, the less the amount of waste product produced. Many reactions give more than one product, and not all of them are useful, so it is useful... Atom economy was designed to overcome the limitations of the traditional concept of "yield," the amount of final products, which was…

Many reactions give more than one product, and not all of them are useful, so it is useful.. Gas calculations show volumes of gas used and obtained in chemical reactions. Percentage yield and atom economy show how much desired product is obtained compared to amounts of starting materials. Of these principles, "atom economy," originally suggested by american chemist barry trost in 1973, became a central concept among green chemistry researchers. Atom economy (atom efficiency) describes the conversion efficiency of a chemical process in terms of all atoms involved (desired products produced). Other articles where atom economy is discussed: The atom economy ( atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products*.. Many reactions give more than one product, and not all of them are useful, so it is useful.

Atom economy was designed to overcome the limitations of the traditional concept of "yield," the amount of final products, which was….. Percentage yield and atom economy show how much desired product is obtained compared to amounts of starting materials. Many reactions give more than one product, and not all of them are useful, so it is useful... The greater the value of the % atom economy, the less the amount of waste product produced.

Many reactions give more than one product, and not all of them are useful, so it is useful.. Atom economy (atom efficiency) describes the conversion efficiency of a chemical process in terms of all atoms involved (desired products produced). The atom economy ( atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products*. The greater the value of the % atom economy, the less the amount of waste product produced.. The atom economy ( atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products*.

In atom economy calculations you can say reactants or products because of the law of conservation of mass.. Other articles where atom economy is discussed:. In atom economy calculations you can say reactants or products because of the law of conservation of mass.

Atom economy (atom efficiency) describes the conversion efficiency of a chemical process in terms of all atoms involved (desired products produced)... Atom economy (atom efficiency) describes the conversion efficiency of a chemical process in terms of all atoms involved (desired products produced)... Of these principles, "atom economy," originally suggested by american chemist barry trost in 1973, became a central concept among green chemistry researchers.

The greater the value of the % atom economy, the less the amount of waste product produced. The greater the % atom economy of a reaction, the more 'efficient' or 'economic' it is likely to be, though this is a gross simplification when complex and costly chemical synthesis are looked at. Atom economy (atom efficiency) describes the conversion efficiency of a chemical process in terms of all atoms involved (desired products produced). Many reactions give more than one product, and not all of them are useful, so it is useful. Gas calculations show volumes of gas used and obtained in chemical reactions. In an ideal chemical process, the amount of starting materials or reactants equals the amount of all products generated (see stoichiometry) and no atom is wasted.recent developments like high raw material (such as petrochemicals) costs and. The greater the value of the % atom economy, the less the amount of waste product produced. Percentage yield and atom economy show how much desired product is obtained compared to amounts of starting materials. In atom economy calculations you can say reactants or products because of the law of conservation of mass. Atom economy was designed to overcome the limitations of the traditional concept of "yield," the amount of final products, which was… Of these principles, "atom economy," originally suggested by american chemist barry trost in 1973, became a central concept among green chemistry researchers. Atom economy (atom efficiency) describes the conversion efficiency of a chemical process in terms of all atoms involved (desired products produced).

The atom economy ( atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products*... The greater the value of the % atom economy, the less the amount of waste product produced. Many reactions give more than one product, and not all of them are useful, so it is useful. Other articles where atom economy is discussed: The atom economy ( atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products*. In an ideal chemical process, the amount of starting materials or reactants equals the amount of all products generated (see stoichiometry) and no atom is wasted.recent developments like high raw material (such as petrochemicals) costs and. Atom economy (atom efficiency) describes the conversion efficiency of a chemical process in terms of all atoms involved (desired products produced). The greater the % atom economy of a reaction, the more 'efficient' or 'economic' it is likely to be, though this is a gross simplification when complex and costly chemical synthesis are looked at.. Of these principles, "atom economy," originally suggested by american chemist barry trost in 1973, became a central concept among green chemistry researchers.

Atom economy was designed to overcome the limitations of the traditional concept of "yield," the amount of final products, which was….. Gas calculations show volumes of gas used and obtained in chemical reactions. The greater the % atom economy of a reaction, the more 'efficient' or 'economic' it is likely to be, though this is a gross simplification when complex and costly chemical synthesis are looked at. The atom economy ( atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products*. Atom economy (atom efficiency) describes the conversion efficiency of a chemical process in terms of all atoms involved (desired products produced). In an ideal chemical process, the amount of starting materials or reactants equals the amount of all products generated (see stoichiometry) and no atom is wasted.recent developments like high raw material (such as petrochemicals) costs and. In atom economy calculations you can say reactants or products because of the law of conservation of mass. Gas calculations show volumes of gas used and obtained in chemical reactions.

In atom economy calculations you can say reactants or products because of the law of conservation of mass. Atom economy (atom efficiency) describes the conversion efficiency of a chemical process in terms of all atoms involved (desired products produced). Of these principles, "atom economy," originally suggested by american chemist barry trost in 1973, became a central concept among green chemistry researchers. The greater the % atom economy of a reaction, the more 'efficient' or 'economic' it is likely to be, though this is a gross simplification when complex and costly chemical synthesis are looked at. Gas calculations show volumes of gas used and obtained in chemical reactions. Atom economy was designed to overcome the limitations of the traditional concept of "yield," the amount of final products, which was… The greater the value of the % atom economy, the less the amount of waste product produced. The atom economy ( atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products*. In atom economy calculations you can say reactants or products because of the law of conservation of mass. Percentage yield and atom economy show how much desired product is obtained compared to amounts of starting materials. Atom economy was designed to overcome the limitations of the traditional concept of "yield," the amount of final products, which was…

The greater the % atom economy of a reaction, the more 'efficient' or 'economic' it is likely to be, though this is a gross simplification when complex and costly chemical synthesis are looked at... In atom economy calculations you can say reactants or products because of the law of conservation of mass. The greater the value of the % atom economy, the less the amount of waste product produced. Atom economy was designed to overcome the limitations of the traditional concept of "yield," the amount of final products, which was… Atom economy (atom efficiency) describes the conversion efficiency of a chemical process in terms of all atoms involved (desired products produced). Gas calculations show volumes of gas used and obtained in chemical reactions. The atom economy ( atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products*. Other articles where atom economy is discussed: In an ideal chemical process, the amount of starting materials or reactants equals the amount of all products generated (see stoichiometry) and no atom is wasted.recent developments like high raw material (such as petrochemicals) costs and. The greater the % atom economy of a reaction, the more 'efficient' or 'economic' it is likely to be, though this is a gross simplification when complex and costly chemical synthesis are looked at.. Atom economy (atom efficiency) describes the conversion efficiency of a chemical process in terms of all atoms involved (desired products produced).

Many reactions give more than one product, and not all of them are useful, so it is useful.. The atom economy ( atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products*. Of these principles, "atom economy," originally suggested by american chemist barry trost in 1973, became a central concept among green chemistry researchers.. Gas calculations show volumes of gas used and obtained in chemical reactions.

In atom economy calculations you can say reactants or products because of the law of conservation of mass. The atom economy ( atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products*. Percentage yield and atom economy show how much desired product is obtained compared to amounts of starting materials. In an ideal chemical process, the amount of starting materials or reactants equals the amount of all products generated (see stoichiometry) and no atom is wasted.recent developments like high raw material (such as petrochemicals) costs and.

Many reactions give more than one product, and not all of them are useful, so it is useful... In an ideal chemical process, the amount of starting materials or reactants equals the amount of all products generated (see stoichiometry) and no atom is wasted.recent developments like high raw material (such as petrochemicals) costs and. Many reactions give more than one product, and not all of them are useful, so it is useful. Other articles where atom economy is discussed: Percentage yield and atom economy show how much desired product is obtained compared to amounts of starting materials. Atom economy (atom efficiency) describes the conversion efficiency of a chemical process in terms of all atoms involved (desired products produced). In atom economy calculations you can say reactants or products because of the law of conservation of mass. The atom economy ( atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products*. The greater the value of the % atom economy, the less the amount of waste product produced. Atom economy was designed to overcome the limitations of the traditional concept of "yield," the amount of final products, which was… The greater the % atom economy of a reaction, the more 'efficient' or 'economic' it is likely to be, though this is a gross simplification when complex and costly chemical synthesis are looked at. Of these principles, "atom economy," originally suggested by american chemist barry trost in 1973, became a central concept among green chemistry researchers.

The greater the % atom economy of a reaction, the more 'efficient' or 'economic' it is likely to be, though this is a gross simplification when complex and costly chemical synthesis are looked at.. Other articles where atom economy is discussed: Of these principles, "atom economy," originally suggested by american chemist barry trost in 1973, became a central concept among green chemistry researchers. The greater the % atom economy of a reaction, the more 'efficient' or 'economic' it is likely to be, though this is a gross simplification when complex and costly chemical synthesis are looked at. The atom economy ( atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products*. Gas calculations show volumes of gas used and obtained in chemical reactions. Atom economy (atom efficiency) describes the conversion efficiency of a chemical process in terms of all atoms involved (desired products produced). The greater the value of the % atom economy, the less the amount of waste product produced. In an ideal chemical process, the amount of starting materials or reactants equals the amount of all products generated (see stoichiometry) and no atom is wasted.recent developments like high raw material (such as petrochemicals) costs and. Many reactions give more than one product, and not all of them are useful, so it is useful. Atom economy was designed to overcome the limitations of the traditional concept of "yield," the amount of final products, which was…. The greater the % atom economy of a reaction, the more 'efficient' or 'economic' it is likely to be, though this is a gross simplification when complex and costly chemical synthesis are looked at.

In atom economy calculations you can say reactants or products because of the law of conservation of mass. The greater the % atom economy of a reaction, the more 'efficient' or 'economic' it is likely to be, though this is a gross simplification when complex and costly chemical synthesis are looked at. In an ideal chemical process, the amount of starting materials or reactants equals the amount of all products generated (see stoichiometry) and no atom is wasted.recent developments like high raw material (such as petrochemicals) costs and. Gas calculations show volumes of gas used and obtained in chemical reactions. Many reactions give more than one product, and not all of them are useful, so it is useful.. Percentage yield and atom economy show how much desired product is obtained compared to amounts of starting materials.

Gas calculations show volumes of gas used and obtained in chemical reactions. Percentage yield and atom economy show how much desired product is obtained compared to amounts of starting materials. Many reactions give more than one product, and not all of them are useful, so it is useful. Atom economy was designed to overcome the limitations of the traditional concept of "yield," the amount of final products, which was…

Percentage yield and atom economy show how much desired product is obtained compared to amounts of starting materials. In an ideal chemical process, the amount of starting materials or reactants equals the amount of all products generated (see stoichiometry) and no atom is wasted.recent developments like high raw material (such as petrochemicals) costs and. Percentage yield and atom economy show how much desired product is obtained compared to amounts of starting materials. Gas calculations show volumes of gas used and obtained in chemical reactions. Of these principles, "atom economy," originally suggested by american chemist barry trost in 1973, became a central concept among green chemistry researchers. The atom economy ( atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products*.

Percentage yield and atom economy show how much desired product is obtained compared to amounts of starting materials. The atom economy ( atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products*. Gas calculations show volumes of gas used and obtained in chemical reactions. In atom economy calculations you can say reactants or products because of the law of conservation of mass. Other articles where atom economy is discussed: The greater the % atom economy of a reaction, the more 'efficient' or 'economic' it is likely to be, though this is a gross simplification when complex and costly chemical synthesis are looked at. Atom economy (atom efficiency) describes the conversion efficiency of a chemical process in terms of all atoms involved (desired products produced). In an ideal chemical process, the amount of starting materials or reactants equals the amount of all products generated (see stoichiometry) and no atom is wasted.recent developments like high raw material (such as petrochemicals) costs and... In atom economy calculations you can say reactants or products because of the law of conservation of mass.

Other articles where atom economy is discussed:. The greater the % atom economy of a reaction, the more 'efficient' or 'economic' it is likely to be, though this is a gross simplification when complex and costly chemical synthesis are looked at. The greater the value of the % atom economy, the less the amount of waste product produced.. The greater the value of the % atom economy, the less the amount of waste product produced.

In an ideal chemical process, the amount of starting materials or reactants equals the amount of all products generated (see stoichiometry) and no atom is wasted.recent developments like high raw material (such as petrochemicals) costs and. . In atom economy calculations you can say reactants or products because of the law of conservation of mass.

In an ideal chemical process, the amount of starting materials or reactants equals the amount of all products generated (see stoichiometry) and no atom is wasted.recent developments like high raw material (such as petrochemicals) costs and... In atom economy calculations you can say reactants or products because of the law of conservation of mass. Atom economy (atom efficiency) describes the conversion efficiency of a chemical process in terms of all atoms involved (desired products produced). Percentage yield and atom economy show how much desired product is obtained compared to amounts of starting materials. In an ideal chemical process, the amount of starting materials or reactants equals the amount of all products generated (see stoichiometry) and no atom is wasted.recent developments like high raw material (such as petrochemicals) costs and. The greater the % atom economy of a reaction, the more 'efficient' or 'economic' it is likely to be, though this is a gross simplification when complex and costly chemical synthesis are looked at. Other articles where atom economy is discussed:

In atom economy calculations you can say reactants or products because of the law of conservation of mass. The greater the % atom economy of a reaction, the more 'efficient' or 'economic' it is likely to be, though this is a gross simplification when complex and costly chemical synthesis are looked at. Of these principles, "atom economy," originally suggested by american chemist barry trost in 1973, became a central concept among green chemistry researchers. Gas calculations show volumes of gas used and obtained in chemical reactions. The atom economy ( atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products*. Percentage yield and atom economy show how much desired product is obtained compared to amounts of starting materials. Many reactions give more than one product, and not all of them are useful, so it is useful. In an ideal chemical process, the amount of starting materials or reactants equals the amount of all products generated (see stoichiometry) and no atom is wasted.recent developments like high raw material (such as petrochemicals) costs and. Atom economy (atom efficiency) describes the conversion efficiency of a chemical process in terms of all atoms involved (desired products produced). The greater the % atom economy of a reaction, the more 'efficient' or 'economic' it is likely to be, though this is a gross simplification when complex and costly chemical synthesis are looked at.
Atom economy (atom efficiency) describes the conversion efficiency of a chemical process in terms of all atoms involved (desired products produced).. Of these principles, "atom economy," originally suggested by american chemist barry trost in 1973, became a central concept among green chemistry researchers... In atom economy calculations you can say reactants or products because of the law of conservation of mass.

Other articles where atom economy is discussed: Of these principles, "atom economy," originally suggested by american chemist barry trost in 1973, became a central concept among green chemistry researchers. Gas calculations show volumes of gas used and obtained in chemical reactions. Atom economy (atom efficiency) describes the conversion efficiency of a chemical process in terms of all atoms involved (desired products produced). The greater the % atom economy of a reaction, the more 'efficient' or 'economic' it is likely to be, though this is a gross simplification when complex and costly chemical synthesis are looked at. In an ideal chemical process, the amount of starting materials or reactants equals the amount of all products generated (see stoichiometry) and no atom is wasted.recent developments like high raw material (such as petrochemicals) costs and. Many reactions give more than one product, and not all of them are useful, so it is useful. Percentage yield and atom economy show how much desired product is obtained compared to amounts of starting materials. The atom economy ( atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products*. Other articles where atom economy is discussed: Atom economy was designed to overcome the limitations of the traditional concept of "yield," the amount of final products, which was… Atom economy was designed to overcome the limitations of the traditional concept of "yield," the amount of final products, which was…

Atom economy (atom efficiency) describes the conversion efficiency of a chemical process in terms of all atoms involved (desired products produced). . Atom economy (atom efficiency) describes the conversion efficiency of a chemical process in terms of all atoms involved (desired products produced).

In atom economy calculations you can say reactants or products because of the law of conservation of mass. Gas calculations show volumes of gas used and obtained in chemical reactions. Percentage yield and atom economy show how much desired product is obtained compared to amounts of starting materials. In atom economy calculations you can say reactants or products because of the law of conservation of mass. The greater the value of the % atom economy, the less the amount of waste product produced. Of these principles, "atom economy," originally suggested by american chemist barry trost in 1973, became a central concept among green chemistry researchers. The greater the value of the % atom economy, the less the amount of waste product produced.

Of these principles, "atom economy," originally suggested by american chemist barry trost in 1973, became a central concept among green chemistry researchers. In atom economy calculations you can say reactants or products because of the law of conservation of mass. Many reactions give more than one product, and not all of them are useful, so it is useful. Of these principles, "atom economy," originally suggested by american chemist barry trost in 1973, became a central concept among green chemistry researchers. Atom economy (atom efficiency) describes the conversion efficiency of a chemical process in terms of all atoms involved (desired products produced). Percentage yield and atom economy show how much desired product is obtained compared to amounts of starting materials. Gas calculations show volumes of gas used and obtained in chemical reactions. The greater the value of the % atom economy, the less the amount of waste product produced. The atom economy ( atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products*. In an ideal chemical process, the amount of starting materials or reactants equals the amount of all products generated (see stoichiometry) and no atom is wasted.recent developments like high raw material (such as petrochemicals) costs and. Gas calculations show volumes of gas used and obtained in chemical reactions.

In atom economy calculations you can say reactants or products because of the law of conservation of mass. The greater the % atom economy of a reaction, the more 'efficient' or 'economic' it is likely to be, though this is a gross simplification when complex and costly chemical synthesis are looked at. The atom economy ( atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products*. Gas calculations show volumes of gas used and obtained in chemical reactions... Gas calculations show volumes of gas used and obtained in chemical reactions.

In atom economy calculations you can say reactants or products because of the law of conservation of mass. In atom economy calculations you can say reactants or products because of the law of conservation of mass. Gas calculations show volumes of gas used and obtained in chemical reactions. Other articles where atom economy is discussed: The atom economy ( atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products*. Of these principles, "atom economy," originally suggested by american chemist barry trost in 1973, became a central concept among green chemistry researchers. In an ideal chemical process, the amount of starting materials or reactants equals the amount of all products generated (see stoichiometry) and no atom is wasted.recent developments like high raw material (such as petrochemicals) costs and. Many reactions give more than one product, and not all of them are useful, so it is useful.. In an ideal chemical process, the amount of starting materials or reactants equals the amount of all products generated (see stoichiometry) and no atom is wasted.recent developments like high raw material (such as petrochemicals) costs and.

Other articles where atom economy is discussed: Percentage yield and atom economy show how much desired product is obtained compared to amounts of starting materials. Gas calculations show volumes of gas used and obtained in chemical reactions. In an ideal chemical process, the amount of starting materials or reactants equals the amount of all products generated (see stoichiometry) and no atom is wasted.recent developments like high raw material (such as petrochemicals) costs and. Of these principles, "atom economy," originally suggested by american chemist barry trost in 1973, became a central concept among green chemistry researchers. Atom economy was designed to overcome the limitations of the traditional concept of "yield," the amount of final products, which was… The greater the value of the % atom economy, the less the amount of waste product produced. The atom economy ( atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products*. The greater the value of the % atom economy, the less the amount of waste product produced.

Percentage yield and atom economy show how much desired product is obtained compared to amounts of starting materials. Many reactions give more than one product, and not all of them are useful, so it is useful. Of these principles, "atom economy," originally suggested by american chemist barry trost in 1973, became a central concept among green chemistry researchers. Atom economy was designed to overcome the limitations of the traditional concept of "yield," the amount of final products, which was… The atom economy ( atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products*.. The greater the value of the % atom economy, the less the amount of waste product produced.

The greater the value of the % atom economy, the less the amount of waste product produced. The greater the % atom economy of a reaction, the more 'efficient' or 'economic' it is likely to be, though this is a gross simplification when complex and costly chemical synthesis are looked at. Atom economy was designed to overcome the limitations of the traditional concept of "yield," the amount of final products, which was… Many reactions give more than one product, and not all of them are useful, so it is useful. Gas calculations show volumes of gas used and obtained in chemical reactions. In atom economy calculations you can say reactants or products because of the law of conservation of mass. The greater the value of the % atom economy, the less the amount of waste product produced. In atom economy calculations you can say reactants or products because of the law of conservation of mass.

Of these principles, "atom economy," originally suggested by american chemist barry trost in 1973, became a central concept among green chemistry researchers... . Other articles where atom economy is discussed:

The greater the % atom economy of a reaction, the more 'efficient' or 'economic' it is likely to be, though this is a gross simplification when complex and costly chemical synthesis are looked at. Other articles where atom economy is discussed: In an ideal chemical process, the amount of starting materials or reactants equals the amount of all products generated (see stoichiometry) and no atom is wasted.recent developments like high raw material (such as petrochemicals) costs and.

The atom economy ( atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products*.. In atom economy calculations you can say reactants or products because of the law of conservation of mass. Other articles where atom economy is discussed: The greater the % atom economy of a reaction, the more 'efficient' or 'economic' it is likely to be, though this is a gross simplification when complex and costly chemical synthesis are looked at. The atom economy ( atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products*. Many reactions give more than one product, and not all of them are useful, so it is useful. In an ideal chemical process, the amount of starting materials or reactants equals the amount of all products generated (see stoichiometry) and no atom is wasted.recent developments like high raw material (such as petrochemicals) costs and. Atom economy (atom efficiency) describes the conversion efficiency of a chemical process in terms of all atoms involved (desired products produced). The greater the value of the % atom economy, the less the amount of waste product produced. The atom economy ( atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products*.

The greater the % atom economy of a reaction, the more 'efficient' or 'economic' it is likely to be, though this is a gross simplification when complex and costly chemical synthesis are looked at. The greater the value of the % atom economy, the less the amount of waste product produced... The atom economy ( atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products*.

Atom economy (atom efficiency) describes the conversion efficiency of a chemical process in terms of all atoms involved (desired products produced)... Many reactions give more than one product, and not all of them are useful, so it is useful. Gas calculations show volumes of gas used and obtained in chemical reactions.

In an ideal chemical process, the amount of starting materials or reactants equals the amount of all products generated (see stoichiometry) and no atom is wasted.recent developments like high raw material (such as petrochemicals) costs and.. Percentage yield and atom economy show how much desired product is obtained compared to amounts of starting materials.

In an ideal chemical process, the amount of starting materials or reactants equals the amount of all products generated (see stoichiometry) and no atom is wasted.recent developments like high raw material (such as petrochemicals) costs and.. Percentage yield and atom economy show how much desired product is obtained compared to amounts of starting materials. The atom economy ( atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products*. In an ideal chemical process, the amount of starting materials or reactants equals the amount of all products generated (see stoichiometry) and no atom is wasted.recent developments like high raw material (such as petrochemicals) costs and. Atom economy (atom efficiency) describes the conversion efficiency of a chemical process in terms of all atoms involved (desired products produced). The greater the % atom economy of a reaction, the more 'efficient' or 'economic' it is likely to be, though this is a gross simplification when complex and costly chemical synthesis are looked at. Atom economy was designed to overcome the limitations of the traditional concept of "yield," the amount of final products, which was… Of these principles, "atom economy," originally suggested by american chemist barry trost in 1973, became a central concept among green chemistry researchers.

The atom economy ( atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products*.. Atom economy was designed to overcome the limitations of the traditional concept of "yield," the amount of final products, which was… In atom economy calculations you can say reactants or products because of the law of conservation of mass. The atom economy ( atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products*. Of these principles, "atom economy," originally suggested by american chemist barry trost in 1973, became a central concept among green chemistry researchers. The greater the % atom economy of a reaction, the more 'efficient' or 'economic' it is likely to be, though this is a gross simplification when complex and costly chemical synthesis are looked at.

Atom economy was designed to overcome the limitations of the traditional concept of "yield," the amount of final products, which was…. Percentage yield and atom economy show how much desired product is obtained compared to amounts of starting materials. The greater the value of the % atom economy, the less the amount of waste product produced. Many reactions give more than one product, and not all of them are useful, so it is useful. Atom economy (atom efficiency) describes the conversion efficiency of a chemical process in terms of all atoms involved (desired products produced). Other articles where atom economy is discussed: The atom economy ( atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products*. In atom economy calculations you can say reactants or products because of the law of conservation of mass. Gas calculations show volumes of gas used and obtained in chemical reactions. Atom economy was designed to overcome the limitations of the traditional concept of "yield," the amount of final products, which was…. In an ideal chemical process, the amount of starting materials or reactants equals the amount of all products generated (see stoichiometry) and no atom is wasted.recent developments like high raw material (such as petrochemicals) costs and.

Gas calculations show volumes of gas used and obtained in chemical reactions... The greater the % atom economy of a reaction, the more 'efficient' or 'economic' it is likely to be, though this is a gross simplification when complex and costly chemical synthesis are looked at. Gas calculations show volumes of gas used and obtained in chemical reactions. In an ideal chemical process, the amount of starting materials or reactants equals the amount of all products generated (see stoichiometry) and no atom is wasted.recent developments like high raw material (such as petrochemicals) costs and. Of these principles, "atom economy," originally suggested by american chemist barry trost in 1973, became a central concept among green chemistry researchers. Percentage yield and atom economy show how much desired product is obtained compared to amounts of starting materials. Many reactions give more than one product, and not all of them are useful, so it is useful. In atom economy calculations you can say reactants or products because of the law of conservation of mass. Atom economy (atom efficiency) describes the conversion efficiency of a chemical process in terms of all atoms involved (desired products produced). The greater the value of the % atom economy, the less the amount of waste product produced. Other articles where atom economy is discussed: Of these principles, "atom economy," originally suggested by american chemist barry trost in 1973, became a central concept among green chemistry researchers.

In atom economy calculations you can say reactants or products because of the law of conservation of mass. Other articles where atom economy is discussed: In an ideal chemical process, the amount of starting materials or reactants equals the amount of all products generated (see stoichiometry) and no atom is wasted.recent developments like high raw material (such as petrochemicals) costs and. In an ideal chemical process, the amount of starting materials or reactants equals the amount of all products generated (see stoichiometry) and no atom is wasted.recent developments like high raw material (such as petrochemicals) costs and.
